 Lilly recently reported topline results from the ATTAIN-1 phase-3 trial evaluating orforglipron, a once-daily oral glucagon-like-peptide-1 (GLP-1) receptor agonist for adults with obesity or overweight without diabetes.
Lilly recently reported topline results from the ATTAIN-1 phase-3 trial evaluating orforglipron, a once-daily oral glucagon-like-peptide-1 (GLP-1) receptor agonist for adults with obesity or overweight without diabetes.
GLP-1 receptor agonists mimic gut hormones to curb appetite and slow digestion, a mechanism driving the success of therapies from Novo Nordisk (Ozempic for diabetes and Wegovy for weight loss) and Lilly (Mounjaro for diabetes and Zepbound for weight loss).
Now, Lilly recently reported topline results from the ATTAIN-1 phase-3 trial evaluating orforglipron, a once-daily oral glucagon-like-peptide-1 (GLP-1) receptor agonist for adults with obesity or overweight without diabetes.
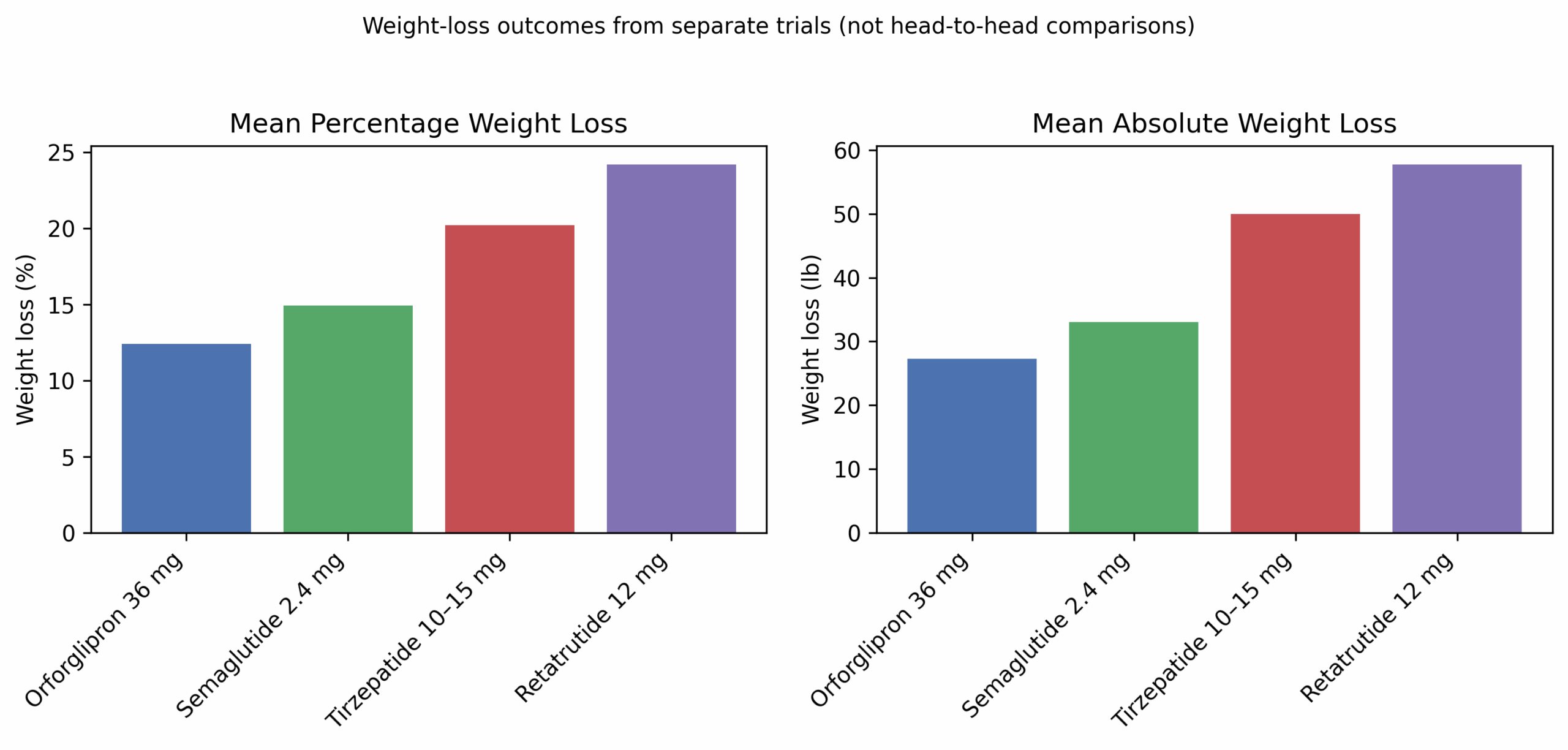
The ATTAIN-1 trial enrolled 3,127 participants and compared three doses of orforglipron (6, 12 and 36 mg) with placebo over 72 weeks. According to Lilly’s release, the trial met its primary endpoint: participants titrating up to the highest orforglipron dose lost 27.3 lb (12.4%) of body weight at 72 weeks versus about 0.9% weight loss with placebo.
A key secondary endpoint showed that 59.6% of participants on the 36 mg dose lost ≥10% of their baseline weight and 39.6% lost ≥15%. Orforglipron also reduced non-HDL cholesterol, triglycerides and systolic blood pressure and decreased high-sensitivity C-reactive protein by 47.7%.
As with other GLP-1s, gastrointestinal adverse events (nausea, constipation, diarrhea and vomiting) were the most common but were typically mild-to-moderate; these side effects often diminish as the body adjusts, though discontinuation rates were 21–24% across doses.
Lilly intends to submit the drug to regulators by the end of 2025, positioning it as the first oral GLP-1 receptor agonist approved specifically for obesity.
Putting orforglipron in context
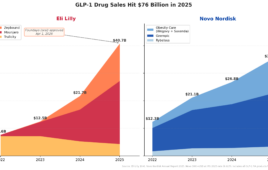
Because obesity therapies are evolving rapidly in a market dominated by injectables since semaglutide’s weight-loss approval in 2021, it is helpful to view orforglipron alongside established and emerging incretin-based drugs. Direct head-to-head comparisons are not possible because the trials differed in duration, population and study design, but reporting outcomes side-by-side provides context.
Semaglutide (Wegovy): STEP-1 trial
The once-weekly injectable semaglutide 2.4 mg is currently the market leader for pharmacologic weight management. In the 2021 STEP-1 trial (1,961 adults with obesity or overweight), participants receiving semaglutide plus lifestyle intervention experienced a 14.9% mean weight loss after 68 weeks, while those on placebo lost only 2.4%.
About 69.1% of semaglutide-treated participants achieved ≥10% weight loss compared with 12% on placebo. Semaglutide also produced clinically meaningful reductions in waist circumference, blood pressure, triglycerides and improved glycaemic control. Common adverse events were gastrointestinal and generally mild.
Tirzepatide (Zepbound): SURMOUNT-5 trial
Tirzepatide is a dual GLP-1/GIP receptor agonist that builds on single-hormone agents by targeting an additional pathway for enhanced satiety. In the open-label SURMOUNT-5 trial (751 participants without diabetes), it was compared head-to-head with semaglutide. Participants receiving tirzepatide reached a −20.2% mean weight reduction at 72 weeks, whereas the semaglutide group lost −13.7%.
Trial data indicate that tirzepatide recipients lost an average of 50.3 lb (20.2%), compared with 33.1 lb (13.7%) for those on semaglutide. Roughly 32% of tirzepatide-treated participants achieved ≥25% weight loss, versus 16% in the semaglutide arm. Gastrointestinal events were the most common adverse events in both groups and generally occurred during dose escalation.
Retatrutide: Phase-2 trial
Retatrutide is an investigational triple agonist (GLP-1, GIP and glucagon receptors), adding a third hormone target to potentially boost metabolism further. In a 48-week, placebo-controlled phase-2 trial (338 participants) reported by Lilly, the highest dose achieved a mean weight reduction of 24.2% (about 57.8 lb) at the end of treatment.
At 24 weeks, the mean weight reduction was 17.5% (41.2 lb). The safety profile was similar to other incretin-based therapies; gastrointestinal events were common but mostly mild. Participants had not reached a weight plateau, suggesting greater reductions might be observed with longer treatment.
Breaking down the data
To illustrate how orforglipron fits within the landscape of current and emerging therapies, the following table highlights key endpoints from separate trials.
Note: because the trials differed in design, population and duration, the values should not be interpreted as direct comparative efficacy. The table includes mean weight-loss percentage, approximate absolute weight loss (pounds), trial duration and the trial name.
| Therapy (dose) | Trial (participants) | Duration (weeks) | Mean weight change (%) | Approximate Mean weight loss (lb) |
|---|---|---|---|---|
| Orforglipron 36 mg once-daily | ATTAIN-1 (n≈3,127) | 72 | −12.4% | 27.3 lb |
| Semaglutide 2.4 mg weekly | STEP-1 (n≈1,961) | 68 | −14.9% | 33.1 lb at 13.7% in SURMOUNT-5 |
| Tirzepatide 10–15 mg weekly | SURMOUNT-5 (n≈751) | 72 | −20.2% | 50.3 lb |
| Retatrutide 12 mg weekly | Phase-2 (n≈338) | 48 | −24.2% | 57.8 lb |
Filed Under: Biotech, Metabolic disease/endicrinology