Sage Therapeutics has pegged the wholesale acquisition price of the oral postpartum depression (PPD) drug Zurzuvae (zuranolone) at $15,900 for a 14-day course of the therapy. The drug, co-developed by Biogen, won the FDA green light in August. The companies plan on launching the drug in December. The DEA has classified zuranolone as Schedule IV,…
How a data lakehouse can give you a panoramic view of your AI-enabled clinical trials
In recent years, the term “data lakehouse” has entered the lexicon of data professionals. For AI-enabled clinical trials, the lakehouse architecture promises seamless integration of diverse data streams, spanning patient health records to real-time sensor data, all processed efficiently and queried in structured formats. The lakehouse architecture aims to provide a comprehensive overview of data,…
An overview of the RSV vaccine landscape: GSK aims to extend its approval of Arexvy?
GSK (NYSE:GSK) is aiming to expand the label for its respiratory syncytial virus (RSV) vaccine Arexvy, which was the first to win FDA approval. The firm is now eyeing an extension of the label to include adults aged 50 to 59, bolstered by encouraging preliminary data from a phase 3 study. The recent data from…
How the latest AI executive order might impact drug development in the U.S.
The White House has released an executive order that contains what it hails as “the most sweeping actions ever taken to protect Americans from the potential risks of AI systems.” Relevant to drug development, a fact sheet on the order describes its aim to help further “the responsible use of AI in healthcare and the…
Quantum leaps in drug discovery? NobleAI joins forces with Microsoft while King’s College London aligns with Kvantify
The word “revolutionize” gets thrown around a lot in describing the potential of emerging technologies. But quantum computing could make good on the promise to disrupt healthcare applications ranging from drug discovery to medical imaging. Theoretically, a quantum computer with 300 quantum bits (qubits) could crunch more calculations in an instant than there are atoms…
A year in review: AI’s evolving role in drug discovery and development in 2023
In the realm of drug discovery, AI is making waves, and 2023 could potentially be a pivotal year for this technology. As the technology enters the popular consciousness, pharma employees are wondering “why they can’t have similar AI-driven tools for their professional tasks,” said Diane Wuest, head of digital R&D at Sanofi, in a recent…
Insilico Medicine taps AI to nominate small molecule inhibitor ISM9274 as a preclinical cancer therapy
Clinical-stage AI company Insilico Medicine has nominated a novel small molecule inhibitor known as ISM9274 as a preclinical candidate for cancer treatment. The company used its PandaOmics AI platform to analyze genomic data from more than 90 tumor types and identified CDK12 as a promising target for multiple cancers including triple-negative breast cancer, lung cancer,…
NLP in drug discovery and the quest for the ‘right’ research elements
In drug discovery and development, data sources are as diverse as they are plentiful. There are comprehensive databases brimming with molecular targets, cellular processes, genomic sequences, proteomic profiles, and metabolite patterns that shed light on disease pathways. Data possibilities in the patient care realm are similarly vast, spanning electronic medical records, imaging datasets, and even…
Mirikizumab shows continued promise for ulcerative colitis after regulatory hiccup
Lilly has announced that its interleukin-23 blocker mirikizumab demonstrated promise in patients with moderately to severely active ulcerative colitis (UC) in the ongoing open-label LUCENT-3 extension study. The trial evaluated two-year efficacy and safety of the monoclonal antibody in patients who had previously undergone UC treatments, including biologics, that were ineffective, stopped working, or were…
Pfizer Ignite: Kathy Fernando’s vision for accelerating biotech innovation
Kathy Fernando, the senior vice president, head of Pfizer Ignite and Pfizer CentreOne, has had a professional trajectory marked by pivotal serendipities. One occurred when attending a seminar at the University of Pennsylvania, where she met Dr. Drew Weissman, a prominent immunologist and RNA vaccine researcher. Weissman, along with Katalin Karikó, recently received the Nobel…
10 major pharma and biotech companies that downsized in 2023
The biopharma industry faced mounting headwinds in 2023, with several major players announcing significant restructuring efforts. Faced with expiring patents, inflation and rising interest rates, and heightened FTC scrutiny, companies looked to trim expenses and refocus their businesses. The ebbing tied of the pandemic posed a challenge for a number of drug developers as well.…
Novo Nordisk stops once-weekly semaglutide kidney outcomes trial early following interim analysis
Novo Nordisk will halt the phase 3b FLOW trial, which investigated the effects of once-weekly injectable semaglutide on kidney outcomes in individuals with type 2 diabetes (T2D) and chronic kidney disease (CKD). The company reached the decision following the recommendation of the independent Data Monitoring Committee (DMC), following an interim analysis that met pre-specified criteria…
Click chemistry breakthroughs drive Shasqi and J&J cancer alliance
In June 2023, the click chemistry-focused startup Shasqi revealed a research pact with Johnson & Johnson Enterprise Innovation. More recently, the company announced that it had expanded the research alliance, furthering work on its intratumorally injected biopolymer, known as SQL70. The collaboration will also apply its clinically validated Click Activated Protodrugs Against Cancer (CAPAC) technology…
Upadacitinib shows promising results in ongoing phase 3 atopic dermatitis studies
AbbVie has shared new efficacy and safety data for Rinvoq (upadacitinib) in adults and adolescents with atopic dermatitis (AD) from a trio of ongoing phase 3 studies. Spanning 140 weeks, these studies sustained the co-primary endpoints of Eczema Area and Severity Index 75 (EASI 75) and validated Investigator’s Global Assessment for Atopic Dermatitis 0/1 (vIGA-AD…
More than a century after its synthesis, MDMA could be headed for FDA approval for PTSD
First synthesized in 1912 by Merck, the empathogenic drug 3,4-Methylenedioxymethamphetamine (MDMA) is inching toward FDA approval following the positive results of a phase 3 study. The recently concluded phase 3 study, MAPP2, published in Nature Medicine, found that MDMA-assisted therapy significantly outperforms traditional talk therapy in reducing PTSD symptoms. Participants receiving MDMA-AT had an 86.5%…
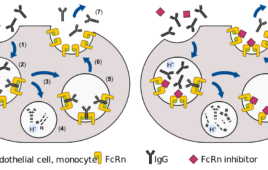
The multi-billion dollar promise of efgartigimod and the broader FcRn inhibitor market
Thanks to promising candidates such as efgartigimod, the Fc receptor (FcRn) inhibitor market is poised to reshape the autoimmune disease treatment landscape. Sales projections for the market top $10 billion, as Driehaus Capital Management estimated in 2020. The addressable U.S. patient base spans more than 228,500 individuals across various conditions including myasthenia gravis, warm autoimmune…
Molecular photoswitches: Illuminating the future of inherited retinal disease treatment
Vision, one of our most valued senses, is a complex orchestration of light-sensing photoreceptors, neurons, and intricate pathways leading to the brain. Central to this native biological system are the photoreceptors – the rods and cones – cells that are responsible for capturing light and initiating visual perception. For many with inherited retinal diseases (IRDs),…
Early data suggest adjunctive cariprazine could be a promising tool for tackling stubborn anhedonia in MDD
The dopamine D3 receptor-targeting cariprazine could potentially be a new tool for managing anhedonia, one of the most stubborn symptoms of major depressive disorder (MDD). Characterized by a lack of enjoyment in previously pleasurable activities, anhedonia has been “difficult to address with traditional antidepressants,” said Dr. Vladimir Maletic, coauthor of a poster presented at the…
Gate Neurosciences takes on depression with event-driven pharmacology
Much of the world is grappling with a mental health crisis — with soaring rates of depression and anxiety. Last year, the startup Gate Neurosciences emerged with a novel approach. While the Carmel, Indiana–based company is focused on synaptic plasticity like many other contemporary CNS companies, it diverges in its strategy. Rather than using a…
Anebulo wins positive FDA feedback to Advance phase 3 program for cannabis
The clinical stage biopharma Anebulo Pharmaceuticals recently shared that it received positive FDA feedback after a Type B meeting in July, where the regulatory agency indicated a path to approval for ANEB-001, a competitive CB1 antagonist with a high affinity for the human CB1 receptor. Overstimulation of this receptor by cannabinoids such as THC causes…
New study sheds light on Eliquis and Xarelto switching
For patients with an elevated stroke risk, switching anticoagulant medications could be a health gamble. Those who change from Eliquis (apixaban) to Xarelto (rivaroxaban) could face almost double the risk of stroke or severe bleeding. Conversely, sticking with Eliquis or transitioning from rivaroxaban to Eliquis appears to be a safer option. Those are key takeaways…
Ketabon GmbH eyes KET01’s potential as an at-home option for treatment-resistant depression
A joint venture between HMNC Brain Health and Develco Pharma in Switzerland, Ketabon GmbH recently released top-line results from its phase 2 KET01-02 study of KET01, a slow-release formulation of oral ketamine, for treatment-resistant depression (TRD). KET01 was associated with improvements in depression severity as early as day 1, but the results were not statistically…
How synthetic data accelerates oncology research and drug development
Synthetic data in oncology is transforming how researchers and developers approach real-world evidence. They often need this evidence to test hypotheses, predict outcomes and develop algorithms. But privacy constraints and access related to patient data can create delays and lengthen project timelines. Oncology drug researchers and developers have recently begun using synthetic data in oncology…
Legacy Healthcare aims to upend alopecia areata treatment with a botanical drug
Imagine a world where botanical drugs could shape the future of medical treatment. Saad Harti, CEO of Swiss company Legacy Healthcare, doesn’t just imagine it; he’s on a mission to make it a reality. With an initial focus on alopecia areata in children and adolescents, Legacy Healthcare believes the plant extract Coacillium could be a…
A checklist for unlocking the promise of AI in clinical trials
AI algorithms offer a myriad of advantages for clinical trials. AI techniques can, for instance, support patient enrollment and site selection, improve data quality and enhance patient outcomes. AI algorithms — combined with an effective digital infrastructure — can also help aggregate and manage clinical trial data in real time, as Deloitte has noted. Last…