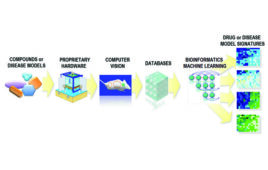
The journey to developing a successful drug, theoretically, may appear linear: you discover the right drug, find the suitable patient and administer it at the right time. The reality, however, often deviates from this straightforward path. Aligning these three variables remains notoriously difficult, often leading to elongated timelines strewn with failures, sometimes extending over a…
Phase 3 trial shows donanemab reduces Alzheimer’s symptoms by 35%
According to results from the Phase 3 TRAILBLAZER-ALZ 2 study published in JAMA, the monoclonal antibody donanemab significantly slowed cognitive and functional decline in patients with early symptomatic Alzheimer’s disease by approximately 35% at one year compared to placebo. The trial enrolled 1736 patients across 277 research centers in 8 countries. Another monoclonal antibody, Leqembi…
QuantHealth taking a data-driven predictive approach to simulate clinical trials
In drug discovery, the process of shooting for regulatory approval can feel less like a sprint and more like a marathon, but with no guarantee of crossing the finish line. Despite the hefty investment of time, effort and resources, the success rate for bringing new drugs to market hasn’t improved in recent decades. The American…
Apitegromab shows sustained benefits in phase 2 SMA trial
Cambridge, Massachusetts–based biopharma Scholar Rock has been making encouraging progress in the development of apitegromab, a potential new therapy for spinal muscular atrophy (SMA), a genetic disease characterized by progressive muscle weakness and atrophy. Apitegromab, a novel monoclonal antibody, works through a unique mechanism of action — it binds to and inhibits the precursor form…
eClinical Solutions Q&A: The quest to transform raw data into drug discovery gold
Top pharmaceutical companies sponsor over a hundred clinical trials annually, generating vast amounts of data. Harnessing this deluge is a monumental task. eClinical Solutions, led by CEO Raj Indupuri, tackles this through advanced applications of data analytics and machine learning with an emphasis on AI in clinical trials optimization. Specifically, eClinical Solutions taps AI/ML for…
Leqembi could mark new era in Alzheimer’s treatment progress: An overview of the evolving drug development scene
Today, the FDA granted traditional approved to lecanemab (branded as Leqembi), a monoclonal antibody from Eisai and Biogen for adult patients with Alzheimer’s disease. The agency made the decision on the basis of a confirmatory trial that showed its clinical benefit. The drug, which reduces the formation of amyloid plaques in the brain, is the…
Seven biologics walking the approval tightrope in 2023
As we venture into the second half of 2023, the global biologics market continues to hover between robust growth and unpredictability. Valued at $461.74 billion in 2022, it’s projected to grow at a compound annual rate of 10.3% from 2023 to 2030, according to Grand View Research. In 2022, biologics edged past small molecules for…
CEO: Insilico on how AI can ‘imagine the perfect molecules’ for drug targets
Insilico Medicine, an AI-based biotech startup, announced details of their first AI-designed drug candidate to enter human clinical trials. INS018_055 is an experimental treatment for idiopathic pulmonary fibrosis, a rare lung disease. Through their Insilico AI-driven drug discovery platform, they discovered and designed INS018_055 in just 30 months, significantly faster than the industry average. Phase…
Insilico’s AI-discovered INS018_055 graduates to phase 2
Roughly a year ago, Insilico Medicine announced that it had dosed the first patient in a phase 1 study of INS018_055, an AI-discovered, first-in-class small molecule inhibitor. Now, the company has progressed to the next stage, launching a phase 2 study for the drug candidate. Insilico, a founding member of NVIDIA Inception, developed its AI…
A deep dive into AWS’s strategy with generative AI and ML in life sciences
The AI market is witnessing meteoric growth, with projections hinting at a potentially staggering increase over the next decade. Against this backdrop of rapid AI evolution, we recently spoke with Tehsin Syed, general manager of AWS Health. Syed shared that Amazon Web Services (AWS) is seeing growing interest from Big Pharma firms. “Nine of the…
FDA publishes draft guidance for psychedelic drug development
In a sign of shifting attitudes, the FDA has released draft guidance to facilitate the development of psychedelic drugs. Historically, psychedelics have a long history of use in certain cultures, such as the indigenous tribes of the Amazon Basin and Native American communities, for millennia as sacraments.These substances were used not only recreationally but also…
Guselkumab could offer hope for psoriatic arthritis patients resistant to TNF inhibitors
Janssen continues to strengthen the case that its interleukin-23 inhibitor TREMFYA (guselkumab) offers promise to many patients with psoriatic arthritis (PsA). The company’s phase 3b COSMOS clinical trial involving 189 patients with active PsA and an inadequate response to one to two previous tumor necrosis factor (TNF) inhibitors, guselkumab showed sustained improvements in measures of…
iBio’s chief reveals strategy behind AI-driven bispecific antibody discovery plans
Biotech firm iBio (NYSEA:IBIO) has incorporated EngageTx, a machine learning-driven technology, into its development roadmap. This T-cell engaging antibody panel assists in generating bispecific antibodies targeting cancer cells. In particular, the firm is developing a novel Trophoblast Cell Surface Antigen 2 (TROP-2) bispecific molecule to target TROP-2-positive cancers. In addition to their focus on oncology,…
AdAPT-001 oncolytic adenovirus shows promising phase 1 cancer treatment results
Oncolytic adenoviruses have won significant attention in recent years as a novel approach to cancer treatment. One example of the trend is AdAPT-001 TGF-ß Trap, an engineered variant of the common cold virus equipped with a transforming growth factor-beta (TGF-β) “trap.” This mechanism is designed to latch onto and neutralize TGF-β, an immunosuppressive cytokine involved…
Beyond the trip with non-hallucinogenic psychoplastogens in neuropsychiatry
Interest in ketamine and psilocybin as potential therapies for mood disorders has surged since around 2010. A groundbreaking 2000 study at Yale revealed the powerful antidepressant effects of ketamine, a dissociative anesthetic. Unlike traditional antidepressants which can take weeks or months to have an impact, a single dose of ketamine led to significant improvements in…
Decoding Bayer’s digital health leap and its implications on drug discovery and personalized medicine
The German multinational pharma and biotech colossus Bayer is taking further steps to ramp up its focus on digital health by launching a new business unit. In 2022, Bayer invested $9.5 million in Woebot Health, an AI-powered behavioral health platform company. In 2020, it launched a venture known as G4A Digital Health Partnership Program to…
Guselkumab offers sustained improvements for psoriatic arthritis patients in phase 3b trial
Data from Janssen’s phase 3b COSMOS clinical trial reveals that guselkumab (Tremfya), an inhibitor of interleukin-23 (IL-23), provides improvements in all minimal disease activity (MDA) domains for adults with active psoriatic arthritis (PsA). The benefits persist through week 48, even in patients who have seen inadequate responses to one or two tumor necrosis factor inhibitors…
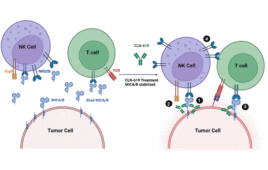
CLN-619 antibody therapy offers a new hope for patients with advanced solid tumors
Boston-based Cullinan Oncology has unveiled data for its new monoclonal antibody therapy, CLN-619, ahead of the American Society of Clinical Oncology (ASCO) 2023 meeting scheduled for June 2–6 in Chicago. The drug could potentially offer a new treatment option for patients with advanced solid tumors. Dr. Judy Wang, Associate Director of Drug Development at the…
Insilico Medicine wins IND approval for AI-designed USP1 inhibitor for cancer trials in U.S. and China
Insilico Medicine has made a significant breakthrough with its AI-designed USP1 inhibitor, ISM3091. The US Food and Drug Administration (FDA) has accepted Insilico’s Investigational New Drug (IND) application for this promising drug, marking a significant milestone for AI-assisted drug discovery. “The FDA’s acceptance of our IND for ISM3091 signifies that the FDA recognizes its potential…
PsychoGenics’ SmartCube prompts a reevaluation of CNS drug discovery
In an era of rapid AI progress, the quest to pioneer the first AI-developed drug candidates has led to an increasing number of these drug candidates entering clinical trials. One contender is ulotaront, an antipsychotic drug, that fared well in a phase 3 schizophrenia study published in NEJM in 2020. Sunovion discovered the drug in…
A current perspective on machine learning’s role in advancing clinical trials diversity
The year 2020 was a watershed moment for many reasons, but notably, it cast a light on the pervasive health and social inequities that have long marred the U.S. The COVID-19 pandemic hit diverse populations disproportionately hard, as Deloitte and others have noted. Additionally, the tragic deaths of George Floyd, Breonna Taylor and others provoked…
Data integrity scandals in biomedical research: Here’s a timeline
While scientific progress hinges on data integrity, plagiarism, data fabrication and image manipulation and other biomedical research scandals are ongoing concerns. An article in Science recently made waves by revealing startling conclusions from the research of the German neuropsychologist Bernhard Sabel, who developed a fake-paper detector. Publishing his findings in a preprint, Sabel concluded that there…
Codagenix taps synthetic biology and machine learning in vaccine development
In the quest to outsmart viral foes such as SARS-CoV-2, RSV and influenza, Codagenix, a clinical-stage biotech firm based in Farmingdale, New York, has engaged a unique arsenal: the intersection of synthetic biology and machine learning. Their weapon of choice is a blend of live-attenuated virus design and codon deoptimization technology. Their process involves introducing…
FDA approves expanded use of Farxiga (dapagliflozin) for heart failure patients regardless of ejection fraction status
FDA has approved an expanded indication for AstraZeneca’s Farxiga (dapagliflozin). This decision, marking a significant milestone in Farxiga’s development, opens up its use in patients with heart failure, regardless of their ejection fraction status. This brings the total number of Farxiga’s approved indications to five. Ejection fraction in heart failure Ejection fraction is a key measure…
Delix Therapeutics launches phase 1 study for novel neuroplasticity-promoting therapeutic
Boston-based Delix Therapeutics has won regulatory approval to commence a study of the non-hallucinogenic psychoplastogen DLX-001. Psychoplastogens are a class of drugs that have the potential to promote neuroplasticity, which refers to the brain’s ability to adapt and form new connections between neurons. That capability could enable improvements in conditions such as depression, anxiety and…