The top 20 healthcare-focused biotech companies collectively raised $2.9 billion in the first quarter of 2024, according to data sourced from Crunchbase. That represents a 161% increase compared to the $1.1 billion raised by the 20 largest funding rounds involving healthcare-focused biotech companies in Q1 2023, indicating more confident bets on the market viability of…
Best-selling pharmaceuticals of 2023 reveal a shift in pharma landscape
Note: This feature on the best-selling pharmaceuticals of 2023 was updated on March 27. Changes are possible as more data become available. Sales of drugs with more than one developer are added together (as in the case of the COVID-19 vaccine Comirnaty) unless one of those companies records the net sales for it, i.e., Dupixent.…
Lantern Pharma aims to take drug to phase 3 for $100-200 million with AI-powered approach
Lantern Pharma’s AI-powered sprint Lantern Pharma (NASDAQ: LTRN), a publicly traded clinical-stage biotech company with a market cap of around $79 million as of mid-March 2024, is shooting for developing $200 million drugs with a machine learning-based platform. The oncology-focused firm Lantern Pharma, profiled last year, has advanced its new drug (LP-284) to a Phase…
Top pharma companies ranked by 2023 R&D spend
Which major pharmaceutical company leads the way in research and development? Data reveals that the top R&D spender in 2023 was Merck & Co., by a wide margin. While Pfizer had almost as much revenue as Merck ($60.1 billion vs. $58.5 billion), its R&D ratio was on the lower end at 18.29%, or $10.7 billion.…
Debiopharm’s multilink technology and partnerships drive oncology pipeline strategy
Debiopharm, an independent Swiss biopharmaceutical company based in Lausanne, seeks to carve a niche in the competitive oncology and infectious disease markets. Its business model focuses on in-licensing promising drug candidates from universities and smaller biotechs, aiming to add value through development. (The company is also partnering with AI-focused firms like VeriSIM Life.) Sandra von…
Devices, disease, and digital: Holy Grail of healthcare AI
In episode 3 of AI Meets Life Sci, DeviceTalks Managing Editor Kayleen Brown and Pharma and Biotech Editor Brian Buntz sit down with GE HealthCare GM of Oncology Ben Newton and Haley Schwartz of Catalyze Healthcare to discuss the impacts of AI to screen, diagnose, prognose, and treat cure disease while addressing real-world implementation issues from regulation and liability to clinician trust and adoption. They review the AI…
Biotech bounces back at JPM 2024 on optimism, breakthroughs and calculated bets, but uncertainties persist
At the dawn of 2024, there’s a sense of renewed optimism in the biotech sector despite recent sector-specific challenges. This week, the JP Morgan Health Care Conference witnessed strong deal-making activity. For instance, Merck agreed to acquire cancer drug developer Harpoon Therapeutics for roughly $680 million, highlighting continued interest in oncology cancer therapies. Meanwhile, Novartis…
What’s next for biotech? Q4 2023 funding trends point beyond the usual suspects
Oncology may continue be one of the hottest sectors across the pharma sector, but other therapeutic areas are catching up in terms of innovation and investment. While oncology and hematology jointly accounted for about one-third of the new FDA approvals in 2023, investors are increasingly betting on precision medicine, advanced drug delivery systems and the use of…
Core trends in 2023 FDA drug approvals: Oncology, neurology and hematology dominate
2023 was a big year for hematology, neurology and oncology, with the medical specialties seeing the most FDA approvals. In terms of sponsors, Pfizer had the most approvals with six total, followed by UCB and Chiesi, each with three apiece. When looking at commercial prospects, AstraZeneca’s respiratory syncytial virus antibody Beyfortus could be the biggest…
Pharma M&A activity primed for another high-flying year in 2024
Despite challenging interest rates and regulatory environment, pharma M&A activity surged in 2023 with deal volume up more than 30% from the prior year based on a review of more than 200 acquisitions since 2018. The total disclosed deal value in 2023 also more than doubled the prior year’s tally to surpass $100 billion. While…
50 of the best-funded biotechs of 2023
As the year draws to a close, it is clear that molecular science and diagnostics is the hottest funding area in the biotech industry. In an analysis of 50 of the best-funded biotechs of 2023 focused on human health, molecular and science and diagnostics startups collectively attracting roughly $945 million, dwarfing the figures in other…
How Lantern Pharma and Code Ocean partnered on oncology drug development
A vision for data-driven drug development in oncology When Peter Carr, principal software architect of Lantern Pharma, stepped into his full-time role in September 2020, the company was on the cusp of a transformation. While AI had been a focus for a number of years, a fresh infusion of cash provided a possibility of expanding…
Nobel-connected startup Shasqi deepens J&J partnership on CAPAC platform
San Francisco-based oncology startup, Shasqi, announced an expansion of its research collaboration with Johnson & Johnson Enterprise Innovation. The partnership centers on Shasqi‘s CAPAC platform, which is an abbreviation for Click-Activated Protodrugs Against Cancer. The platform separates tumor-targeting from the actual drug payload with the aim of maximizing potency while minimizing toxic side effects. Shasqi’s…
Johnson & Johnson pharma rebrand highlights innovation as a pillar to reinforce trust
Global pharma and medical device giant Johnson & Johnson (J&J) has ditched its iconic cursive logo that dates back to the late 19th century, and rebranded its Janssen pharma division as Johnson & Johnson Innovative Medicine. The move underscores the company’s push to prioritize higher-margin prescription drugs. This strategic move comes amidst a backdrop of…
Mapping the cancer patient journey with liquid biopsy
According to the American Cancer Society, one in two men and one in three women will be diagnosed with cancer at some point in their lives. Patients seek treatment to shrink their tumors and ideally achieve remission; however, there is no one-size-fits-all approach. At its core, cancer is a genetic disease: Different types of cancer that affect…
iBio’s chief reveals strategy behind AI-driven bispecific antibody discovery plans
Biotech firm iBio (NYSEA:IBIO) has incorporated EngageTx, a machine learning-driven technology, into its development roadmap. This T-cell engaging antibody panel assists in generating bispecific antibodies targeting cancer cells. In particular, the firm is developing a novel Trophoblast Cell Surface Antigen 2 (TROP-2) bispecific molecule to target TROP-2-positive cancers. In addition to their focus on oncology,…
The 50 best-selling pharmaceuticals of 2022: COVID-19 vaccines poised to take a step back
The COVID-19 pandemic has had a profound impact on the best-selling pharmaceuticals, leading to shifts in the list with Pfizer and BioNTech’s Comirnaty surpassing AbbVie’s Humira for the No. 1 spot in 2021. That momentum continued in 2022, with Pfizer and BioNTech jointly raking in $59.1 billion in revenue from the sales of the COVID-19…
Understanding cellular heterogeneity and its implications for disease diagnosis and treatment
Cellular heterogeneity enables cells in the body to perform an array of functions, but it also plays a role in diseases like cancer as well. In oncology, this fact influences molecular therapy targets, tumor evolution as well as drug sensitivity and resistance. Cellular heterogeneity is also a consideration in areas such as immunotherapy, neuroscience, stem…
Prominent pharma M&A deals to watch in Q1 2023: Pfizer, Sanofi and others on the hunt
After a relatively quiet 2022, 2023 is anticipated to be a more robust year for pharma M&A activity. In recent months, players such as Pfizer (NYSE:PFE), Sanofi (NYSE:SNY), Chiesi, AstraZeneca (LON:AZN) and Amgen (Nasdaq:AMGN) have unveiled acquisition plans that could reshape the landscape of the sector. This article delves into the details of prominent recent…
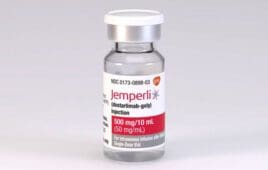
Dostarlimab drug trials: The science behind cancer and the therapy’s future
Cancer treatment has been a critical point for clinical diagnostics. Over the years, various treatment approaches- from naked nucleic acid-based therapy, targeting microRNAs, oncolytic virotherapy, and suicide-gene-based therapy to CRISPR/Cas-9-based therapy. More recently, drug-based therapy has also emerged as a promising area for cancer therapeutics. Regarding drug administration, a major buzz has been created around…
Why research on hematologic malignancies is ramping up
Oncology has benefited from a wave of advances in recent decades. From 1991 to 2018, the age-adjusted overall cancer death rate dropped 31%, according to an analysis from American Association for Cancer Research (AACR). That death rate dropped 2.4% between 2017 and 2018, marking the most significant annual reduction. The reasons for such improvements are multifaceted,…
How Mission Bio is working to drive precision medicine
Mission Bio (San Francisco) is a growth-stage company spun out of the microfluidics lab at the University of California San Francisco (UCSF) in 2014. “We have been commercial since late 2017,” said the company’s CEO, Yan Zhang, in a recent interview. Initially focusing on oncology, Mission Bio has cast its gaze across precision medicine to…
Black Diamond Therapeutics slashes workforce by 30% while discontinuing development of BDTX-189
Precision oncology firm Black Diamond Therapeutics (Nasdaq:BDTX) has announced a restructuring plan to free up cash to prioritize the development of BDTX-1535 and BDTX-4933. The company plans on trimming nearly one-third of its workforce. BDTX shares ticked up about 1% to $2.75 after announcing the news yesterday but fell 4% today to $2.64. According to…
GSK to purchase biopharma Sierra Oncology for $1.9 billion
GlaxoSmithKline plc (LSE/NYSE:GSK) has reached a deal to acquire Sierra Oncology (Nasdaq:SRRA) to bolster its oncology pipeline. Under the terms of the proposed deal, GSK would pay $44 per share for a total value of approximately $1.9 billion. GSK shares were mostly flat in mid-day trading, dipping 0.032% to $46.52. SRRA shares surged 38.31% to…
President Biden reboots Cancer Moonshot project
In 2016, then-Vice President Joe Biden announced a Cancer Moonshot initiative to speed the development of new therapies to treat cancer. Biden is now launching a new initiative committed to improving the cancer survival rate by at least 50% by 2047. The plan also aims to improve the quality of life for patients with cancer.…