The year 2020 was a watershed moment for many reasons, but notably, it cast a light on the pervasive health and social inequities that have long marred the U.S. The COVID-19 pandemic hit diverse populations disproportionately hard, as Deloitte and others have noted. Additionally, the tragic deaths of George Floyd, Breonna Taylor and others provoked…
Data integrity scandals in biomedical research: Here’s a timeline
While scientific progress hinges on data integrity, plagiarism, data fabrication and image manipulation and other biomedical research scandals are ongoing concerns. An article in Science recently made waves by revealing startling conclusions from the research of the German neuropsychologist Bernhard Sabel, who developed a fake-paper detector. Publishing his findings in a preprint, Sabel concluded that there…
Codagenix taps synthetic biology and machine learning in vaccine development
In the quest to outsmart viral foes such as SARS-CoV-2, RSV and influenza, Codagenix, a clinical-stage biotech firm based in Farmingdale, New York, has engaged a unique arsenal: the intersection of synthetic biology and machine learning. Their weapon of choice is a blend of live-attenuated virus design and codon deoptimization technology. Their process involves introducing…
FDA approves expanded use of Farxiga (dapagliflozin) for heart failure patients regardless of ejection fraction status
FDA has approved an expanded indication for AstraZeneca’s Farxiga (dapagliflozin). This decision, marking a significant milestone in Farxiga’s development, opens up its use in patients with heart failure, regardless of their ejection fraction status. This brings the total number of Farxiga’s approved indications to five. Ejection fraction in heart failure Ejection fraction is a key measure…
Delix Therapeutics launches phase 1 study for novel neuroplasticity-promoting therapeutic
Boston-based Delix Therapeutics has won regulatory approval to commence a study of the non-hallucinogenic psychoplastogen DLX-001. Psychoplastogens are a class of drugs that have the potential to promote neuroplasticity, which refers to the brain’s ability to adapt and form new connections between neurons. That capability could enable improvements in conditions such as depression, anxiety and…
FDA places clinical hold on Sun Pharma’s dermatology drug as a result of blood clot risk
Sun Pharmaceuticals (NSE:SUNPHARMA) has received a clinical hold from the FDA on its experimental dermatological drug deuruxolitinib over the potential for thromboembolic incidents. The hold pertains to patients taking a 12-mg dose of the Janus kinase (JAK) inhibitor. Reuters was the first to report the news. Sun Pharma maintains confidence in deuruxolitinib despite FDA clinical…
Discover the 25 top pharma employers of 2023
The pharma and biotech sectors continue to face rapid change. On one hand, top pharma employers have continued to face talent shortages as demand increases for experienced workers. But on the other, the pharma sector has not been immune to layoffs. One factor driving the trend is the increased clip of mergers and acquisitions (M&A)…
The future of rapid and enduring neuropsychiatric treatments: From psychedelics to non-hallucinatory psychoplastogens
Neuropsychiatric disorders, such as depression and anxiety, are among the leading causes of disability worldwide. Almost one billion people globally — roughly one out of eight individuals — live with a mental disorder, with anxiety and depressive disorders the most common, according to the World Health Organization. Yet current therapies such as selective serotonin reuptake…
Unraveling the impact of FDORA and PREVENT Pandemics Acts on the life sciences
As the world continues to grapple with global health challenges, the role of science and biotech law has taken center stage in shaping public health policy and innovation. The FDORA and PREVENT Pandemics Acts are poised to help shape the landscape. In a recent interview, life sciences attorney Barbara Binzak Blumenfeld offers insights into significant…
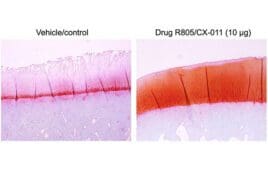
Q&A: USC researchers uncover potential osteoarthritis treatment breakthrough with R805/CX-011
Researchers at the Keck School of Medicine at the University of Southern California (USC) have discovered a drug compound, R805/CX-011, with the potential to alleviate the painful hyperinflammation from osteoarthritis. The findings concerning the prospective osteoarthritis treatment breakthrough, published in Science Translational Medicine, showed positive effects in animal models and could pave the way for…
Brain breakthroughs: Aprinoia Therapeutics’ harnesses AI and strategic partnerships to propel neurodegenerative disease research
Neurodegenerative disease research is witnessing significant advances. To that end, Hong Kong-headquartered Aprinoia Therapeutics is embracing a ‘precision neuroscience’ approach to neurodegeneration diagnostics. The company’s lead program, APN-1607, represents a new generation of advanced tau positron emission tomography (PET) tracers, which play a crucial role in effective and efficient diagnosis of patients suffering from neurodegenerative…
HELIOS trial Q&A: Amylyx Pharmaceuticals’ AMX0035 as a potential treatment for Wolfram syndrome
The recently launched phase 2 HELIOS clinical trial from Amylyx Pharmaceuticals, aims to explore the potential of AMX0035 (sodium phenylbutyrate and taurursodiol) in treating Wolfram syndrome, a rare and complex genetic disorder. In a recent interview, Drs. Lahar Mehta and Fumihiko Urano discussed the AMX0035 Wolfram syndrome trial design, objectives and implications for future research…
Three strikes in pharma: Exploring recent drug withdrawals and clinical trial challenges
Pharmaceutical companies face a long list of regulatory challenges ranging from patent expiry to bioequivalence and international harmonization. It’s not uncommon for drug makers to withdraw or discontinue drugs after failing to meet clinical requirements or endpoints, resulting in drug withdrawals. On average, life science companies pull roughly 4,500 drugs and devices from the market,…
Landscape overview: Moderna and Pfizer lead the race in mRNA flu vaccines, with plans for regulatory filings in 2024-2025
mRNA technology helped propel the development of some of the most successful drugs in pharma history — notably, the Comirnaty COVID-19 from Pfizer/BioNTech generated almost $56 billion in 2022 while Moderna’s Spikevax vaccine raked in $18.4 billion. But demand for COVID-19 vaccines continues to cool. In 2023, Comirnaty brought in $15 billion between its two…
Breaking down barriers: Prioritizing diversity in clinical trials
Diversity in clinical trials is crucial for ensuring that new medical treatments are safe and effective for all populations. Last year, the FDA released draft guidance that aims to improve the enrollment of historically underrepresented populations in clinical trials. Demand is growing for more transparency and diversity in clinical trials, according to Erin Leckrone, senior…
Dostarlimab shows promise in improving endometrial cancer treatment outcomes
Endometrial cancer, a common gynecological malignancy, leaves considerable unmet needs for patients with advanced or recurrent disease. GSK‘s (NYSE:GSK) Jemperli (dostarlimab), a PD-1 receptor antagonist, brings hope to the numerous women diagnosed with this cancer each year. As the first immunotherapy approved for patients with deficient mismatch repair (dMMR) endometrial cancer, dostarlimab has the potential…
Best practices for data capture in long-term follow-up studies
Clinical trials and long-term follow-up studies (LTFUs) hinge on a whirlwind of data collection, where the volume and quality of patient data form the cornerstone of success. Erratic and patchy data can throw a monkey wrench into evaluating the potency and safety of cutting-edge drugs and devices. Such snags can drag approvals through the mud…
How EEG and machine learning are transforming epilepsy clinical trials
Epilepsy is a brain disorder that triggers recurring seizures. It is the fourth the most common neurological disorders in the world, according to the Epilepsy Foundation. The Centers for Disease Control and Prevention estimates that 65 million people worldwide have active epilepsy. In 2015, 1.2% of the total U.S. population — 3 million adults and…
Exploring the future of mRNA therapy: Where are we now?
Interest in mRNA therapy has exploded in recent years following the development of mRNA-based COVID-19 vaccines, but can the platform live up to the initial hype that surrounded it? More companies have announced clinical trial data from mRNA trials for conditions ranging from cardiovascular disease to cancer. mRNA, or messenger RNA, is a type of…
Breaking through the status quo: Moving beyond traditional safety signal detection
Signal detection is an essential pharmacovigilance (PV) activity designed to support patient safety by actively uncovering potential safety risks in medical drugs and vaccines. For post-market products, the status quo for signal detection involves monitoring individual case safety reports (ICSR) and literature from multiple sources. Since the turn of the century, organizations have relied on…
Scleroderma treatment could leap forward with FT011 drug candidate
The story of the scleroderma drug candidate FT011 is one of “tenacity and persistence,” said Darren Kelly, CEO and managing director of Certa Therapeutics, which is developing the drug. The drug was discovered around 2006 by Kelly’s laboratory at the University of Melbourne and Spencer Williams of the Bio21 Institute. “It was really [Williams] being the…
Neuron23 launches clinical trial for NEU-723 in Parkinson’s with companion diagnostic in development
South San Francisco–based Neuron23 has started the first-in-human Phase 1 trial of NEU-723 for the treatment of Parkinson’s disease. NEU-723 is a highly potent and selective leucine-rich repeat kinase 2 (LRRK2) inhibitor. This clinical trial will evaluate the safety, tolerability and pharmacokinetics of NEU-723 in healthy volunteers. A targeted approach The company has also collaborated…
Community oncology groups driving more clinical trials
In the U.S., most cancer patients receive care in a community setting, a unique care model that traces its origins back to the original war on cancer. Since then, the National Cancer Institute and other organizations have supported this model of pushing more cancer care to community settings. Community oncology groups are poised to conduct a…
Big Pharma companies trimming pipelines in 2023
Several companies, including GSK, Janssen, Novartis and Pfizer, have announced that they are cutting their pipelines as they release their full-year 2022 financial results. The following are the drugs removed from these companies’ pipelines. Bristol Myers Squibb Anti-CTLA-4 NF: The oncology monoclonal antibody candidate directed against the human T-cell receptor cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) known as BMS-986218 was…
Moderna’s RSV vaccine mRNA-1345 was more than 80% effective in a Phase 3 study
mRNA vaccine company Moderna (Nasdaq:MRNA) plans on submitting for regulatory approval for its investigational RSV mRNA-1345 vaccine after it met primary efficacy endpoints in the Phase 3 ConquerRSV study (NCT05127434). The RSV data are “a clear positive for MRNA” and represent a good omen for upcoming mRNA Phase 3 influenza data, noted UBS in a…