 Cancer diagnoses and screenings dramatically dropped during the peak of the COVID-19 pandemic.
Cancer diagnoses and screenings dramatically dropped during the peak of the COVID-19 pandemic.
Recent data from tens of thousands of patients suggest that the U.S. is likely to see at least two years of increased cancer diagnosis rates and poorer outcomes, according to Jeff Elton, CEO of ConcertAI.
EHR data reflect a pronounced drop in pan tumor diagnoses and multiple myeloma (MM) diagnoses.
Against that backdrop, Elton recommends that the oncology field adopt a data-driven approach to optimize patient care in the wake of the pandemic.
On one level, the dip in diagnostic rates in the early days of the pandemic was not surprising, given how common lockdowns were in early 2021. At that time, it seemed likely that the situation would play a role in driving later-stage oncology diagnosis. The problem is especially evident in the hematological malignancy population.
“The early phase of the pandemic — before vaccination — really hit people with hematological malignancies exceptionally hard,” Elton said.
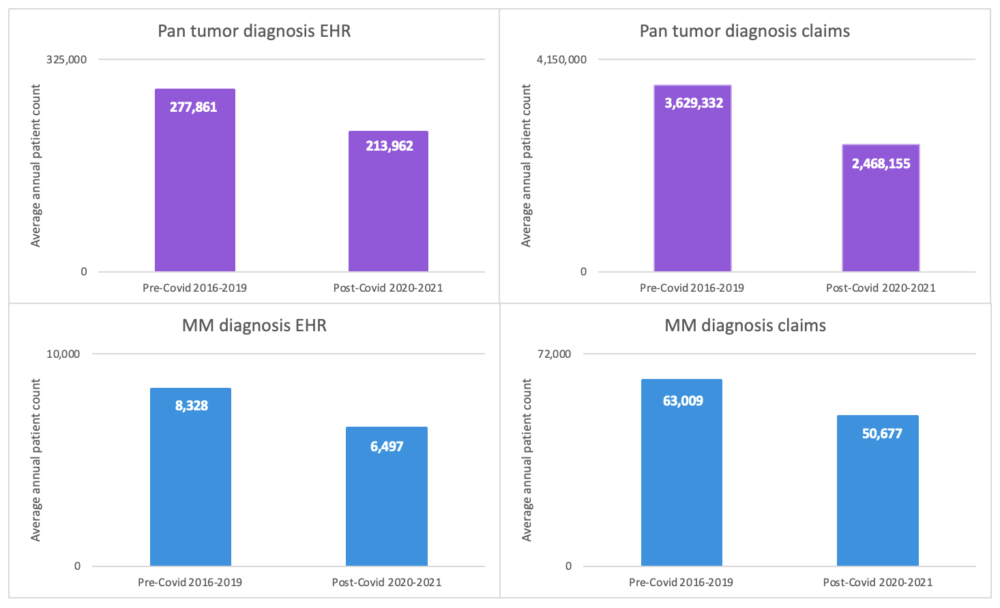
[Image courtesy of Concert.ai]
The pandemic may temporarily reverse that trend, although “we don’t really know what the mortality implications are,” Elton said.
Generally speaking, there’s an expectation that the dip in screening rates during the pandemic will be negative. “If I can diagnose earlier, my treatment options are greater,” Elton said.
And diagnosing cancer at an earlier stage could lead to dramatically improved outcomes. For melanoma, for instance, a Stage I–II diagnosis was associated with a 97.6% 5-year relative survival rate. By contrast, a stage III diagnosis had a 60.3% 5-year survival rate, while the corresponding figure for a stage IV diagnosis was 16.2%.
Elton said the pandemic’s tendency to lead to later-stage cancer diagnosis could impact oncology clinical trials.
“We may want to think about [recruiting cancer patients] slightly earlier than we would normally because these people could be much more progressed in their disease than they would have been normally,” he explained. “We may want to think about a more aggressive intervention in a cancer that has a higher likelihood of having a positive impact rather than just going to the traditional standard of care.”
Lagging cancer screen rates also have implications for real-world data, including for clinical development and research. For example, the provider community may need to approach cancer surveillance slightly differently than in the past. “They likely will be seeing a larger proportion of slightly sicker and more-progressed patients than they would have historically been involved with,” Elton said.

Jeff Elton [Image courtesy of ConcertAI]
“We’re now seeing radiomics features that can predict genomic mutation that’s driving a particular tumor,” Elton said.
The pandemic has also played a role in driving integration in international oncology data sharing while supporting more flexible treatment options for patients. COVID-19 played a role in reducing, for instance, some of “the onerousness of European privacy rules to allowing data sharing to take place for research and quality of care purposes,” Elton said.
The increased openness in data sharing is poised to support a more mature data-driven approach in oncology in the years to come.
Additionally, while the field of oncology tends to require in-patient care, the pandemic played a role in liberalizing telehealth and telemedicine, which can allow some cancer patients to scale back in-patient visits.
The increasing maturity of the data-driven approach in oncology and greater use of telehealth for some degree of routine care are not likely to retreat. “Very rarely do you take away things in healthcare once you have started to establish utility,” Elton said.
Filed Under: clinical trials, Drug Discovery, Oncology



