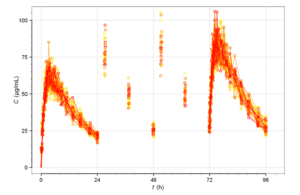
Time course of drug plasma concentrations over 96 hours, following oral administrations every 24 hours. Image courtesy of Wikipedia.
Drug formulation involves balancing the body’s ability to use and then eliminate the drug candidate. The field of pharmacology is dedicated to this balancing act, studying how a drug candidate influences biological systems and the body’s reaction to that compound. This task involves the study of pharmacokinetics (PK) and pharmacodynamics (PD).
Pharmacokinetics is the study of the movement of compounds within the body involves absorption, distribution, metabolism, and excretion (ADME) testing. ADME testing applies to pharmaceutical compounds, including food additives, cosmetics and pesticides.
In pharma, the first part of the ADME process, “absorption,” involves studying how a drug candidate travels from its administration site into the bloodstream. “Distribution” follows the compound’s dispersion to organs after administration, including the level of the compound traveling to target organs. “Metabolism” refers to the initial compound’s disintegration into metabolites and the kidneys’ potential to excrete them. Finally, “excretion” involves studying the body’s ability to rid itself of the compound and metabolites. Compounds may be expelled through a range of potential exit points, including urine, feces, the lungs, skin and breast milk.
PK models can require periodic reassessment and reevaluation.
Pharmacodynamics refers to the study of a drug’s effect on the body. The discipline involves assessing the biochemical and physiological effects of a drug at various doses. The process includes studying a compound’s therapeutic benefits and toxicity. In addition, pharmacodynamics is the study of the affinity, potency and efficacy (here, referring to the response of a drug).
Pharmacodynamics is multidimensional, involving studying a drug’s effects at molecular, biochemical, and physiologic levels.
Assessing a compound’s potential benefits and toxicity requires an assessment of available therapies for the target indication and the severity of the disease in question.
Filed Under: Drug Discovery and Development





Tell Us What You Think!
You must be logged in to post a comment.