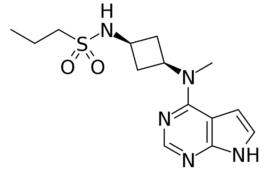
Sanofi (NSE:SANOFI) and its partner GSK (NYSE:GSK) are hoping the low price of their experimental COVID-19 vaccine will stoke interest. In a call with analysts, Thomas Triomphe, head of the Sanofi Pasteur vaccines division, said that its vaccine would cost less than $10 per dose. For the sake of comparison, Pfizer-BioNTech and Moderna COVID-19 vaccines generally cost…
Sanofi completes Translate Bio acquisition
Sanofi (NSDQ:SNY) announced today that it has completed the purchase of its mRNA partner Translate Bio. The previously announced deal, according to Sanofi, accelerates the French pharma giant’s efforts to develop transformative vaccines and therapies using mRNA technology. The deal comes during a year in which mRNA-based Moderna (NSDQ:MRNA) and Pfizer-BioNTech (NYSE:PFE/NSDQ:BNTX) COVID-19 vaccines are…
Pfizer’s abrocitinib goes head-to-head with Sanofi’s Dupixent
Pfizer (NYSE: PFE) recently announced that its once-daily oral Janus kinase 1 (JAK1) inhibitor abrocitinib bested Sanofi’s Dupixent (dupilumab) in a Phase 3 study focused on moderate to severe atopic dermatitis (AD). Meanwhile, Sanofi (EPA:SAN) announced that a Dupixent pivotal trial met its primary and secondary endpoints, making it the first biologic to demonstrate significant…
FDA approves Sanofi’s Nexviazyme for late-onset Pompe disease
With its recent approval of Nexviazyme (avalglucosidase alfa-ngpt), Sanofi (NSDQ:SNY) has asserted its control over the Pompe disease landscape. In 2006, FDA approved Genzyme’s Myozyme (alglucosidase alfa) for Pompe disease. Sanofi acquired the company a decade ago. Additionally, FDA approved Lumizyme in 2010, which uses the same biologic, but is indicated for patients eight years of…
Sanofi reshapes mRNA landscape with $3.2B Translate Bio buy
Sanofi (NSDQ:SNY) may be one of the biggest vaccine manufacturers in the world, but the quick rise of the mRNA vaccine platform during the pandemic caught the company by surprise. The company could be on the way to rectify that with its $3.2 billion acquisition of its mRNA partner Translate Bio. Sanofi began working with…
Several companies could vie for next-gen COVID-19 vaccine market share
A couple of years ago, mRNA vaccines remained at the research stage. Now, they are among the best-selling pharmaceutical products with Pfizer (NYSE:PFE), BioNTech (NSDQ:BNTX) and Moderna (NSDQ:MRNA) dominating the market. However, that could change in the long run as a growing number of companies develop next-generation mRNA COVID-19 vaccines, which promise to be more effective…
Sanofi to spend nearly half a billion dollars a year on mRNA vaccine research
Sanofi (NYSE:SNY) announced today that it plans to invest approximately $476.3 million (€400 million) per year on mRNA vaccine development. Paris-based Sanofi’s intends for its annual investment to go toward a first-of-its-kind vaccine “mRNA Center of Excellence” to accelerate the development and delivery of next-generation vaccines. Approximately 400 employees will integrate end-to-end mRNA vaccine capabilities…
Sanofi and GSK start Phase 3 trial of COVID-19 vaccine
Sanofi and GlaxoSmithKline announced today that they began enrollment in the Phase 3 clinical trial for their COVID-19 vaccine candidate. The companies will evaluate the safety, efficacy and immunogenicity of their adjuvanted recombinant-protein COVID-19 vaccine candidate in a global, randomized, double-blind, placebo-controlled Phase 3 study of more than 35,000 participants aged 18 and older, according…
Pharma 50: Here’s how the world’s largest pharma companies are doing
The global pharmaceutical industry held up well during the pandemic, with 10 of the largest businesses only seeing a roughly –3% drop in revenue in 2020. Eight of the 10 even came out ahead. That’s one of the big takeaways from Drug Discovery & Development’s inaugural Pharma 50, a compilation of data on the largest…
GSK, Sanofi launch new study for protein-based COVID-19 vaccine
Sanofi (NYSE:SNY) and Glaxo Smith Kline (NYSE:GSK) are moving forward with their development plans of a COVID-19 vaccine. In December, the two companies announced that their vaccine development efforts had been delayed after the product failed to mount a robust immune response in older adults. The two companies believe the setback resulted from an insufficient…
Launch of Sanofi and GSK COVID-19 vaccine could be delayed until late 2021
Sanofi (NYSE:SNY) and Glaxo Smith Kline (NYSE:GSK) have confirmed a delay in their adjuvanted recombinant protein-based COVID-19 vaccine owing to an inadequate immune response in older adults in a Phase 1/2 trial. Sanofi and GSK now plan to launch a Phase 2b study in February 2021 to investigate a new vaccine formulation. If that study reaches primary endpoints,…
Sanofi, GSK offer 20M COVID-19 vaccine doses to COVAX alliance
Sanofi (NSDQ:SNY) and GSK (LON: GSK), which have teamed up to produce a COVID-19 vaccine candidate, have signaled their intent to work with the legal administrator of the COVAX Facility. The two companies have vowed to produce 200 million doses of a vaccine based on the protein subunit AS03 adjuvant. Governments, businesses and health organizations…
Sanofi to launch COVID-19 vaccine trial in Q4
Sanofi (NYSE:SNY) and Translate Bio (NSDQ:TBIO) announced today that a Phase 1/2 clinical trial for its COVID-19 vaccine candidate is set for the fourth quarter of 2020. Preclinical results for MRT5500, the COVID-19 vaccine candidate under development by the two companies, demonstrated a favorable immune response profile against SARS-CoV-2, the virus causing coronavirus, which supports the selection…
What are the top COVID-19 vaccine candidates?
Five COVID-19 vaccine candidates are going through Phase 3 clinical trials to prove safety and efficacy. Researchers worldwide are testing 132 COVID-19 vaccine candidates, including 42 that are in clinical trials on humans and at least 92 in preclinical or animal trials, according to the New York Times. With the U.S. alone topping 200,000 COVID-19…
Canada lining up millions of doses of COVID-19 vaccines
Sanofi (NYSE:SNY), GlaxoSmithKline (NYSE:GSK) and Moderna (NSDQ:MRNA) have all agreed to supply Canada with a total of more than 90 million doses of a COVID-19 vaccine. Beginning in 2021, Government of Canada is set to receive up to 72 million doses of an adjuvanted COVID-19 vaccine from Sanofi and GSK, while the country also increased its confirmed…
Drug companies agree not to release COVID-19 vaccine until it’s ready
Executives from nine different drug companies today announced a pledge to ensure safety with a potential COVID-19 vaccine. The CEOs of AstraZeneca (NYSE:AZN), BioNTech (NSDQ:BNTX), GlaxoSmithKline (NYSE:GSK), Johnson & Johnson (NYSE:JNJ), Merck (NYSE:MRK), Moderna (NSDQ:MRNA), Novavax (NSDQ:NVAX), Pfizer (NYSE:PFE) and Sanofi (NYSE:SNY), all of whom are developing vaccine candidates, all signed the pledge, which vows to “uphold the integrity of the scientific process” as…
Sanofi to acquire MS drug-development partner for $3.68B
Sanofi (NYSE:SNY) announced today that it agreed to acquire all outstanding shares of Principia Biopharma (NSDQ:PRNB) in a deal worth approximately $3.68 billion. The deal sets all outstanding shares of Principia at $100 per share, which will be acquired in cash. Sanofi and Principia both received unanimous approval from their respective boards of directors, according to…
Sanofi, GSK to supply up to 60 million doses of COVID-19 vaccine to UK
Sanofi (NYSE:SNY) and GlaxoSmithKline (NYSE:GSK) announced that they reached an agreement with the UK government to supply up to 60 million doses of a COVID-19 vaccine. Subject to a final contract, the UK government will receive the doses of a vaccine candidate developed by the two companies in partnership and based on the recombinant protein-based technology Sanofi used to…
Sanofi, GSK seek to have an adjuvanted COVID-19 vaccine ready by late 2021
Sanofi and GSK recently announced that they have signed a letter of intent to collaborate on an adjuvanted vaccine for COVID-19 — with a goal of having the vaccine available by the second half of 2021. Sanofi is contributing its S-protein COVID-19 antigen. The company’s recombinant DNA technology produced an exact genetic match to proteins…
FDA clears Verily Study Watch for additional capabilities
Verily, Google’s life science’s sister company under Alphabet (NSDQ:GOOGL), has received an FDA 510(k) clearance to include an irregular pulse monitor in its Study Watch wearable. Pharmaceutical industry giants including Novartis, Otsuka, Pfizer and Sanofi formed a strategic alliance with Verily last year to develop patient-centered clinical research programs using Verily’s Project Baseline evidence generation platform…
Sanofi eyes Synthrox purchase
Sanofi (NYSE:SNY) announced today that it is planning to tender an offer to acquire all outstanding common stock shares of clinical-stage biotech company Synthorx. The agreement and plan of merger between the two companies was announced on Dec. 7 and the offer is being made pursuant to that. Sanofi said it stands to acquire the common…