The top 20 healthcare-focused biotech companies collectively raised $2.9 billion in the first quarter of 2024, according to data sourced from Crunchbase. That represents a 161% increase compared to the $1.1 billion raised by the 20 largest funding rounds involving healthcare-focused biotech companies in Q1 2023, indicating more confident bets on the market viability of…
Unleashing a new frontier: The power of germline clinico-genomic data to drive therapeutic development
Over the past decade, the use of deeper sources of real-world data across all stages of the drug development life cycle has become increasingly important to guide disease understanding, trial designs, clinical guidelines, regulatory submissions and post-market studies. The advent of these deeper sources was prompted by the HITECH Act, which had the effect of…
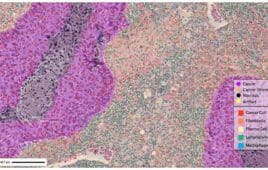
Inside the AI-powered Roche-PathAI companion diagnostics collaboration
PathAI and Roche Tissue Diagnostics (RTD) have inked an exclusive collaboration to develop AI-enabled companion diagnostics that builds on their initial partnership announced in October 2021. To date, PathAI and Roche have commercially launched four algorithms through the partnership. The new partnership will provide biopharma sponsors with integrated technology for developing companion diagnostics incorporating AI-based…
Pitt’s high-performance computing upgrade signals accelerated translational research
The University of Pittsburgh’s recent, significant expansion of its high-performance computing capabilities, courtesy of a gift from Dell Technologies, indicates a strategic commitment to using large-scale data analysis for faster translational research. The additional 9.672 gigaFLOPS of computational power – translating to nearly 9.7 trillion additional computations per second – could help Pitt’s Innovation Hub…
Denmark teams up with Novo Nordisk Foundation, NVIDIA to launch visionary AI research center
A collaboration between the Novo Nordisk Foundation, the Export and Investment Fund of Denmark (EIFO), and NVIDIA will establish a national AI Innovation Centre in Denmark focused on accelerating research and innovation in fields including healthcare, life science, and quantum computing. The initiative is led on the Danish side by the Novo Nordisk Foundation, which…
Why Merck shelled out $30B+ on R&D in 2023
In 2023, Merck & Co. made a considerable investment in research and development, spending more than $30 billion. This figure represents more than double the company’s R&D spending ($13.5 billion) in 2022. The closest competitor in terms of R&D investment was Roche Pharmaceuticals, with a 2023 R&D expenditure of roughly $14.7 billion. This sum is…
Pushing the frontier of drug discovery with the world’s most powerful supercomputer
Oak Ridge National Laboratory’s Frontier, the world’s first exascale supercomputer with its dizzying 1.1 exaflop speed, is a game-changer for scientific domains ranging from drug discovery to material science and oceanography. The computer holds the top spot on the TOP500 list, an independent ranking of the world’s most powerful supercomputers. “It’s like having a million…
Off with the training wheels: AI-based patient characterization can improve clinical trial performance without large data sets
Only 12% of new drug candidates that enter phase 1 clinical development ultimately receive FDA approval. This dismal success rate leaves millions of patients with unmet medical needs and drives up the costs for the small number of drugs that make it to market. More frustratingly, it leaves untold numbers of potentially transformative therapies back-burnered…
How digital pathology can transform the value of clinical trial information
The field of pathology is gradually going digital, marking a pronounced shift from the traditional use of glass slides, a practice that dates back to the Victorian era. Digital pathology uptake reached an “exponential” pace in the wake of the pandemic, noted Dr. Monika Lamba Saini, pathologist-scientist, director of digital innovation at Q2 Solutions, an…
Deepcell’s CEO forges her own path to transforming cell science with AI and microfluidics
Deepcell co-founder Maddison Masaeli took an unexpected path from academic research to launching an AI-powered cell analysis platform. The company has raised close to $100 million in funding to support its mission. This includes a $73 million Series B round and an earlier $20 million Series A round. The company has entered into a research…
Merck, Amgen back Culmination Bio’s quest to transform healthcare data analytics
In the same week that Merck and Amgen revealed expanded alliances with AWS, the bioinformatics startup Culmination Bio revealed that it has received $10 million in funding from the venture arms of those companies, Merck Global Health Innovation Fund and Amgen Ventures. Culmination Bio, a spinoff from Intermountain Health, has developed a vast data lake…
50 of the best-funded biotechs of 2023
As the year draws to a close, it is clear that molecular science and diagnostics is the hottest funding area in the biotech industry. In an analysis of 50 of the best-funded biotechs of 2023 focused on human health, molecular and science and diagnostics startups collectively attracting roughly $945 million, dwarfing the figures in other…
Alzheimer’s at an inflection point as drug and diagnostics breakthroughs emerge
Alzheimer’s disease research appears to be hitting its stride, thanks to recent therapeutic advances in drug development and the emergence of biomarkers to detect the condition. “All the pieces of the puzzle of precision medicine, which is already quite common in oncology, are now in place,” said Hartmuth Kolb, vice president, neuroscience biomarkers and R&D…
PathAI launches AISight digital pathology platform and NSCLC algorithm
PathAI has launched a new digital pathology platform, AISight. Supporting AI-driven research, the digital pathology platform was trained on a real-world dataset of over 5,000 samples with inputs from more than 350,000 cell and tissue-level annotations from more than 50 pathologists. The company has also released its AIM-PD-L1 NSCLC RUO algorithm, which calculates the percentage…
The transformation of precision medicine in infectious disease
Before Alexander Fleming discovered penicillin, another more selective antibacterial agent rose to popularity in the early 1900s: bacteriophage. In 1917, microbiologist Felix d’Herelle was tasked with identifying the cause of a dysentery outbreak impacting French troops. From his research, he noticed that Shigella bacteria was the primary culprit of this affliction. He then discovered an…
How precision drug-dosing supports individualized treatment
The concept of precision drug-dosing has gained ground in recent years, given its ability to boost efficacy and curb side effects. Yet imprecise dosing regimens continue to be common for many drugs, leading to significant rates of adverse drug reactions (ADRs). “ADRs are one of the top ten causes of death in the developed world,”…