Eli Lilly (NYSE:LLY) announced today that it entered into a definitive agreement to acquire Versanis, a private, clinical stage biopharmaceutical company. Versanis focuses on the development of new medicines for the treatment of cardiometabolic diseases. Its lead asset, bimagumab, is a monoclonal antibody. Bimagumab binds activin type II A and B receptors to block activin…
Lilly completes acquisition of Akouos
Eli Lilly and Company (NYSE:LLY) announced today that it successfully completed its acquisition of Akouos (Nasdaq:AKUS). Indianpolis-based Eli Lilly’s acquisition expands its efforts in genetic medicines. Akouos offers a portfolio of potential first-in-class adeno-associated viral gene therapies. The gene therapies treat inner ear conditions, including sensorineural hearing loss. The company initially announced the acquisition in…
7 diabetes treatment innovations to look out for on World Diabetes Day
World Diabetes Day — Nov. 14 — centers around raising awareness for those with diabetes. This year, that aim remains the same, and medical technology companies continue to look for ways to continue improving the management of the metabolic disease. Some of those treatments involve insulin, which was discovered as a treatment for diabetes in…
How Lilly Oncology is aiming to improve clinical trial diversity
Minorities tend to participate in clinical trials at far lower rates than their real-world demographics and prevalence of the disease, according to the American Society of Clinical Oncology. In addition, minorities often have worse outcomes for certain cancers than the broader public. Just one example: the mortality rate for Black women with breast cancer is 40% higher than…
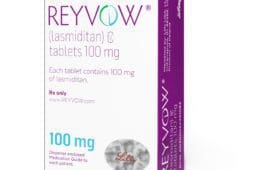
Reyvow indicated for treating migraines
Reyvow (lasmiditan) is an FDA-approved treatment from Lilly for the acute treatment of migraine with or without aura in adults. FDA has not approved it as a preventive treatment of migraine. In October, Lilly announced that the drug showed superior pain freedom after two hours of ingestion in two-thirds of migraine attacks in a Phase…
Eli Lilly acquires glucose-sensing insulin developer Protomer Technologies
Eli Lilly (NYSE:LLY) announced today that it acquired peptide- and protein-engineering platform developer Protomer Technologies. The potential value of the transaction is over $1 billion, depending on the achievement of future development and commercial milestones. Pasadena, Calif.–based Protomer Technologies develops next-generation protein therapeutics designed to identify and synthesize molecules that can sense glucose or other endogenous…
Lilly’s tirzepatide bests semaglutide in type 2 diabetes trial
Eli Lilly (NYSE:LLY) has announced that its investigational drug tirzepatide led to more substantial blood glucose and body weight improvements in a Phase 3 trial than semaglutide, a diabetes drug from Novo Nordisk (NYSE:NVO) that recently scored FDA approval for weight loss in early June. Lilly’s SURPASS-2 results published in The New England Journal of Medicine show tirzepatide…
HHS halts distribution of Lilly’s bamlanivimab/etesevimab antibody cocktail
U.S. government authorities have decided to immediately pause the distribution of Eli Lilly’s bamlanivimab and etesevimab over concerns that the monoclonal antibody cocktail is ineffective against some SARS-CoV-2 variants. CDC recently concluded that the Gamma variant (P.1) and the Beta variant (B.1.351) make up 11% of COVID-19 infections. They are also continuing to become more…
Lilly’s push for accelerated FDA approval of Alzheimer’s drug donanemab
Eli Lilly (NYSE:LLY) has received breakthrough therapy designation from FDA for the investigational antibody donanemab for Alzheimer’s disease. The company plans to file a biologics license application for the drug under the accelerated approval pathway later this year. Like Biogen’s (NSDQ:BIIB) and Eisai’s (TYO:4523) recently approved aducanumab, donanemab is a monoclonal antibody clearing sticky amyloid plaques.…
FDA authorizes lower dose of REGEN-COV COVID-19 antibody cocktail
Regeneron (NSDQ:REGN) has announced that the FDA has signed off on a 1,200 mg subcutaneous or intravenous dose of its REGEN-COV antibody cocktail. The quantity is half of the initially authorized dose. The agency had previously authorized a 2,400 mg dose of the vaccine, including a combined dose of 1,200 mg of Casirivimab and 1,200…
Lilly’s bamlanivimab and etesevimab cut COVID-19 hospitalization and deaths in study
A recent Phase 3 trial found that Lilly’s bamlanivimab (LY-CoV555) and etesevimab (LY-CoV016) administered jointly led to an 87% reduction in hospitalization and deaths in high-risk patients recently diagnosed with COVID-19. The randomized, double-blind, placebo-controlled trial administered 700 mg of bamlanivimab (LY-CoV555) and 1400 mg of etesevimab (LY-CoV016) to 511 patients in the treatment arm.…
FDA authorizes Eli Lilly’s monoclonal antibody treatment for COVID-19
The FDA announced that it issued emergency use authorization (EUA) for monoclonal antibodies developed by Eli Lilly (NYSE:LLY) for treating COVID-19. Eli Lilly’s bamlanivimab and etesevimab received authorization to be administered together for treating mild to moderate COVID-19 in adults and pediatric patients over 12 years old and weighing at least 40 kilograms (about 88 pounds),…
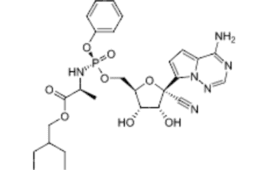
3 notable types of innovative drugs from 2020
Last year, FDA approved 53 drugs, leading the industry to describe 2020 as “a strong year for new drug therapy.” There are several drugs that stand out, according to Todd Wills, the co-author of a study that analyzes how innovative drugs are based on their structure. The drugs that follow are examples of notable innovative therapies.…
11 COVID-19 therapies you need to know
The pharmaceutical industry launched a herculean effort to battle the COVID-19 pandemic early in 2020, developing an array of vaccines and other therapeutics while also identifying existing drugs suitable for treating patients sick with severe coronavirus infections. FDA recently granted emergency authorization to two SARS-CoV-2 vaccines and several other treatments. Here are 11 of the…
Lilly and UnitedHealth team up on bamlanivimab study for COVID-19
Eli Lilly (NYSE:LLY) and UnitedHealth Group (NYSE:UNH) are partnering on a pragmatic study of bamlanivimab (LY-CoV555) in high-risk patients with COVID-19 infections. Lilly recently won emergency use authorization from the FDA to use the monoclonal antibody bamlanivimab to treat mild to moderate COVID-19 infections in patients at risk of severe infections. The study will evaluate the…
Eli Lilly wins FDA nod for COVID-19 antibody treatment
The FDA issued an emergency use authorization (EUA) yesterday for the investigational monoclonal antibody therapy bamlanivimab (LY-CoV555) to treat COVID-19. The drug from Eli Lilly (NYSE:LLY) is intended to reduce the risk of severe COVID-19 infections when administered within 10 days of developing symptoms. The manufacturer indicates the drug for mild-to-moderate COVID-19 infections in adult…
Trump administration paying Eli Lilly $375M for experimental COVID-19 therapy
U.S. HHS and DoD will pay $375 million to buy 300,000 doses of bamlanivimab 700 mg from Eli Lilly and Co. (NYSE:LLY) as part of Operation Warp Speed initiatives to stockpile experimental COVID-19 treatments for distribution, pending FDA authorization. HHS plans to allocate 300,000 doses of the monoclonal antibody immediately, according to an announcement from…
NIAID halts Lilly COVID-19 antibody study
An arm of NIH has ended a clinical trial investigating the use of Eli Lilly’s (NYSE:LLY) monoclonal antibody LY-CoV555 (bamlanivimab) to treat hospitalized COVID-19 patients. In separate news, HHS plans to allocate 300,000 doses of the monoclonal antibody immediately, according to an announcement. While the drug shows promise in treating mild-to-moderate COVID-19 infections, the ACTIV-3 trial…
Report: Eli Lilly halts COVID-19 treatment trial
Eli Lilly (NYSE:LLY) reportedly paused a clinical trial testing its COVID-19 antibody treatment candidate because of a “potential safety concern.” The New York Times reported that Eli Lilly’s testing site researchers were notified of the pause by emails sent by government officials (it is a government-sponsored trial) and the company later confirmed it. A spokesperson from…
The most innovative pharmaceutical agents of 2020
The Galien Foundation recently announced nominees for most innovative pharmaceutical agents for its 14th Annual Prix Galien USA Awards. The foundation bestows the Prix Galien Award annually to examples of outstanding biomedical, pharmaceutical drugs and medical technology products that are designed to improve the human condition. “As we celebrate 50 years of the Prix Galien…