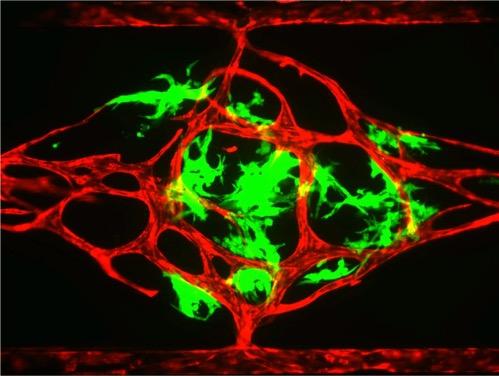
This is an image of a vascularized microtumor (VMT) within a chamber unit of a 96 well plate. Blood vessels are stained in red, and tumor cells in green. Source: Hughes Lab
Led by UCI professor of molecular biology & biochemistry Christopher C.W. Hughes, the research team successfully established multiple vascularized micro-organs on an industry-standard 96-well plate. Hughes and the study’s first author, Duc T. T. Phan, showed that these miniature tissues are much better at reproducing human drug responses than previous model systems. Hughes and his group have shown how the flow of a blood substitute through the vascular network they created can deliver nutrients to multiple kinds of tissues, including heart, pancreas, brain and various tumors.
“This is truly a unique platform – we have recreated in a dish the key element common to all tissues, which is that they depend on blood vessels for their survival. This feature is missing in all previously described in vitro organ cultures,” Hughes said.
Hughes’ team was also able to establish a functional vascularized microtumor (VMT) within the 96-well plate system and demonstrated its potential for anti-cancer drug screening. Working with a panel of FDA-approved anti-cancer drugs and a human colon cancer, they found that the VMT platform could accurately identify drugs that target the tumor cells, the vessels that supply them, or both.
“This is a major breakthrough”, continued Dr. Hughes, “For the first time we can identify in the same assay drugs that target both tumor cells and the vessels that feed them.”
The full study can be found online in the Royal Society of Chemistry’s journal Lab on a Chip.
Filed Under: Drug Discovery




