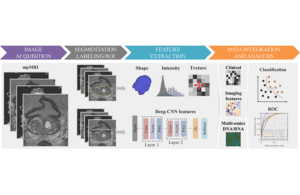
Radiomics image courtesy of Wikimedia Commons
Radiomics, or the science of advanced imaging analytics, is an emerging field that promises more personalized care, improved clinical decision support and greater efficiency in clinical trials.
Radiomics are being used to discover new biomarkers, improve the accuracy of diagnosis, predict the risk of disease and likelihood of treatment response, and identify clinical endpoints.
Today, much of the focus in radiomics has been in oncology. However, radiomics applies to other clinical areas and is being used today in several clinical domains. Radiomic data are a key component of a “multi-omics” approach to medicine, in which radiomic data is combined with data sets from other sources such as genomic and clinical data. Adding more robust phenotype data to these data fabric will present clinicians with synergistic information in which the whole is greater than the sum of its parts, leading to more precise insights required in treatment planning and therapy response.
Following is a quick background on radiomics, a look at how the science is in use today and a glimpse of how clinicians may leverage radiomics to improve patient care in the future.
Radiomics 101
While electronic health records draw much of the popular attention, EHRs are just one source of healthcare data. Indeed, 90% of all healthcare data comes from medical imaging, and more than 97% of that data goes unanalyzed or unused.
Part of the reason that so much of that data is unused lies in radiologists’ traditional methods of reviewing or “reading” medical images, such as CT, MRI or PET scans. Generally, when radiologists review an image of a tumor or lesion, their view is limited to two dimensions – length and width, also known as “the long and short.” To determine how and whether potentially cancerous tumors and lesions are growing or progressing, radiologists use calipers to measure changes across these two axes – which is akin to viewing an overhead photo of an iceberg. However, most of the real action is occurring below the surface.
That is because tumors and lesions – whether cancerous or not – exhibit multiple dimensions in addition to numerous other defining structural features. Different types of cancers, for example, show significantly different phenotypic characteristics that can be noninvasively extracted using AI applied to standard medical images. The resulting data and associated patterns yield highly specific insights or biomarkers.
Radiomics unleashes powerful insights about the characteristics and progress of lesions and tumors that are not discernible from traditional image-reading methods. At its foundation, radiomics starts by building an artificial-intelligence-enabled model based on thousands of healthy organ images. The algorithms ingest thousands of variations from that model that show lesions, tumors, their progress, and outcomes. As a result, rather than being limited to looking simply at length and width, these AI-driven analytics can use this information to extract unseen data from the digital image to measure and determine depth, volume, density, doubling the size, tumor textures and other features that cannot be observed optically in an image.
How researchers are using radiomics today
Today, researchers across the globe are taking advantage of radiomics’ ability to extract data from digital images to gain new insights and advance patient care. Here are three examples:
Identify inclusion and exclusion criteria for clinical trials: Researchers from H. Lee Moffitt Cancer Center & Research Institute performed a study of sarcoma patients and concluded that radiomics was a useful tool for establishing inclusion and exclusion criteria for patient enrichment in clinical trials. Researchers performed a retrospective study on patients from a clinical trial to determine whether radiomic analysis of a particular subset of patients would have produced a different endpoint outcome. The researchers concluded that one radiomic feature, Short Run Emphasis, was the most informative for establishing criteria. Had radiomic analysis been used for inclusion, the trial would have met its primary survival objective.
Predict therapy response: An article published in Cancers illustrated radiomics’ ability to identify biomarkers that predict whether immunotherapy could help certain patients with lung adenocarcinoma. Researchers used radiomic technology to analyze biomarkers to predict patients’ overall survival and progression-free survival time. They concluded that specific radiomic features can be used to select patients with lung adenocarcinoma who would benefit from immunotherapy.
Improve clinical decision-making: In a 2021 publishing in Cancer Control, researchers leveraged radiomics to evaluate chest CT scans of lung-carcinoma patients, demonstrating that the technology decreased variability and showed efficiency gains when compared with analysis conducted by radiologists. In addition, researchers concluded that radiomics metrics could be used to track changes in patients’ tumor sizes over time, data that clinicians could use to assess or predict the response to cancer treatment in the lungs.
The future: Oncology and beyond
While much of the application of radiomics has focused on its benefits for oncology, other clinical areas where there is an opportunity to analyze lesion and structural changes could be improved through the use of the science, including cardiology, neurology, and hepatology. Ophthalmology, in particular patients experiencing diabetic retinopathy, is an area in which radiomics could offer several advantages in the analysis of therapy response.
Nonetheless, we will continue to see expanded use of radiomics across the cancer journey from early identification and risk stratification to treatment planning, monitoring disease progression and response to therapy. Importantly, to gain the watershed moment for clinical use and adoption, the application of radiomics must be automated as much as possible, presenting information in a clinician-friendly way that doesn’t require extra effort or disruption to workflows.

Rose Higgins
Finally, it will be critical to ensure that key stakeholders are part of the radiomics conversation so that payers and policy are aligned to see the benefit and value to the patient.
Rose Higgins serves as Chief Executive Officer of HealthMyne, a pioneer in applied radiomics, the cutting-edge field of extracting novel data and biomarkers from medical images.
Filed Under: clinical trials, Drug Discovery





Tell Us What You Think!
You must be logged in to post a comment.