
[Image courtesy of WuXi AppTec]
Ten oligo-based drugs have been approved since 2018, with 16 approved drugs in total. Additionally, more than 100 Phase 2 or Phase 3 clinical trials are currently underway. However, many unique challenges remain for oligo drugs. The novelty, complexity and evolving regulatory guidance of these therapies can pose greater technical challenges than conventional drugs. In this article, we will focus on how to overcome the challenges of metabolite identification (MetID) and profiling.
What are oligos?
Oligonucleotides are synthetic single or double strands of modified RNA (ribonucleic acid) or DNA (deoxyribonucleic acid). They are less than 100 nucleotides (Nt) and can be used to modulate gene expression by binding to RNA or DNA through the Watson-Crick base pairing principle.
In theory, oligos can target any gene of interest because only the nucleotide sequence of interest along the targeted DNA or RNA needs to be selected. This characteristic makes oligos an exciting therapeutic option within precision medicine.
There are four types of oligos, but all share the properties of being negatively charged and hydrophilic, although they range in molecular weight:
- Antisense oligonucleotides (ASOs)
- Single strand DNA or RNA: 5k-10kDa (~18-30 nucleotides)
- Small interfering RNAs (siRNA)
- Double strand RNA: 14k~17kDa (~20-25 nucleotides for each strand)
- Aptamers
- Single strand DNA or RNA: ~6k-15kDa (~20-100 nucleotides)
- Micro RNAs (miRNA):
- Single strand RNA: 5K-10kDa (~20-25 nucleotides)
With the opportunity for novel and targeted treatment comes unique challenges in delivery, stability and specificity. The action sites for oligos lay in the intracellular space, so they must overcome several biological barriers to reach their pharmacological targets in vivo. These barriers include the following:
- Structural instability
- Easy degradation by nuclease in vivo
- Negatively charged molecular structures
- Difficulty in penetrating cell membranes
- Low escape efficiency from endocytosomes to cytoplasm
- Immunogenicity with the human immune system
Overcome the sample analysis challenges of oligos during MetID
Recent advancements in chemical modification and delivery systems have allowed drug developers to overcome many of the aforementioned challenges of oligos with novel and targeted treatments. Since oligo therapies are intended to alter a biological target, drug developers must be able to accurately determine oligo concentration in biological samples. However, challenges remain for the bioanalysis of oligos.
These characteristics of oligos also present significant challenges for sample analysis. Due to their vulnerable nature, oligos require special assays and handling methods to overcome those challenges in the processes of sample treatment and LC-MS analysis, including the following:
- Readily degraded by nucleases
- Protein and nonspecific binding
- Low recovery of extraction
- Poor chromatographic retention
- Multiple charge states
- Ion suppression
Specific experimental materials, reagents and methods were developed for sample treatment, e.g., liquid-liquid extraction and solid-liquid extraction, which could result in acceptable recovery and stability.
As oligos are highly polar compounds, they are difficult to obtain retention and good peak shape by conventional reversed-phase chromatography. Due to this challenge, ion-pair reversed-phase liquid chromatography (IP-RPLC), hydrophilic interaction chromatography (HILIC), and ion exchange chromatography (IEC) are typically used instead. The most popular method is ion-pair reversed-phase chromatography, which mainly uses triethylamine (TEA), N, N-diisopropylethylamine (DIPEA), triethylamine acetate (TEAA) and hexafluoroisopropanol (HFIP) to form ion-pair reagents.
The study systems of oligos during MetID
It is essential to understand oligo drug metabolism when identifying metabolites that are formed during in vitro and in vivo studies. Metabolites have the potential to be pharmacologically active and could cause off‐target toxicity. Furthermore, the mechanisms of oligos are quite different from conventional metabolic pathways that are described for small-molecule drugs. Identifying the metabolites in early studies may help researchers determine if there is potential for off‐target toxicity, which is important when interpreting the results of preclinical and clinical studies. The tissue distribution of these compounds is also of interest.
Cross-species MetID can help compare metabolic profiles to find if there are unique metabolites to humans in vitro and to support animal species selection for toxicology testing. By comparing the in vitro metabolic data with that from in vivo animals, the correlation of in vitro and in vivo metabolism can be speculated.
Unlike small-molecule drugs, which are mainly metabolized by cytochrome P450-mediated mechanisms, oligos are metabolized mainly by nuclease-mediated cleavage of phosphodiester bonds. Cleavage at the 3-terminus by exonucleases is the major metabolic fate for oligonucleotides, followed by 5-exonuclease and endonuclease cleavage events. Commonly, plasma, S9, and tissue homogenate are used as the in vitro metabolism models to evaluate the metabolism of oligos. Preclinical in vivo MetID studies involved plasma, urine, bile, feces and tissues from animals to evaluate metabolism and clearance pathways in animals, which will facilitate quantitative bioanalysis of any relevant metabolites, including chain-shortened metabolites.
Application of high-resolution mass spectrometry for oligo MetID and profiling
Developments in high-resolution mass spectrometry (HRMS) have contributed to the resurgence of oligo therapy. Oligo metabolites produced by exo- and endo-nuclease activity have been identified using mass spectrometry with accurate mass measurement; however, data interpretation is difficult and time-intensive. This is mainly due to the large number of fragment ions generated from oligos, which can largely be mitigated through improvements and advances in software such as automated spectrum deconvolution and data analysis.
Negative ion ESI-MS has been used for oligo detection and analysis due to the charged phosphodiester backbone. Ion-pairing (IP) reversed-phase LC has been used for the analysis of oligonucleotides, which could overcome the challenge of the high polarity and hydrophilicity that lead to poor retention on conventional reversed-phase chromatography and could be compatible with MS-based techniques. Although ion-pairing reagents lead to ion suppression on mass spectrometry, oligo-dedicated HRMS enables highly sensitive and accurate detection that distinguishes mass difference within 10 ppm, making it possible to identify metabolites of oligos.
HRMS is well-suited to profile and identify oligo metabolites, however, many drug developers do not have the resources and experience to do so in-house. Drug developers should seek out a high-quality laboratory testing partner to assist them with bioanalysis for oligo metabolites.
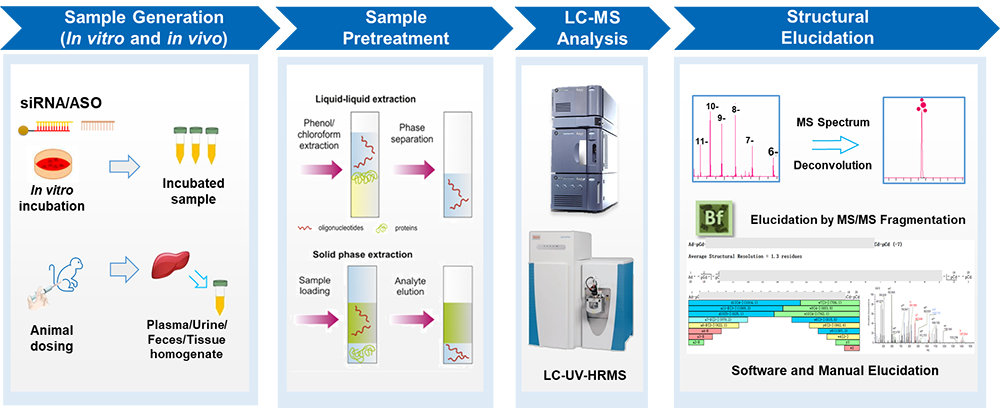
Figure 1. Platform for whole process of oligonucleotide MetID (siRNA and ASO as example) [Image courtesy of WuXi AppTec]
A final word on oligonucleotides
Inherent properties related to oligos, such as instability and negative charge, can pose significant technical challenges to develop novel and targeted oligo therapies; those challenges remain for oligo MetID and profiling studies. HRMS enables accurate detection of ion fragments, thereby making it possible to identify oligo metabolites. Drug developers should seek out a laboratory testing partner that has extensive experience with metabolite testing for oligo drugs. The ability to do so could greatly accelerate oligo therapy development.
References
- Lara Moumné, et al., Oligonucleotide Therapeutics: From Discovery and Development to Patentability. Pharmaceutics 2022, 14, 260.
- Łukasz Nuckowski, et al., Review on Sample Preparation Methods for Oligonucleotides Analysis by Liquid Chromatography. Journal of Chromatography B 2018, 1090, 90-100.
- Ju Liu, et al., Oligonucleotide quantification and metabolite profiling by high-resolution and accurate mass spectrometry. Bioanalysis (2019) 11(21), 1967–1981.
Authors
Gengyao Qin, Principal Scientist in DMPK Service Department of WuXi AppTec, has over 13 years of research experience in preclinical and clinical metabolite profiling and identification of drugs. She has successfully supported over 100 new drugs for IND filing globally.
Dr. Liqi Shi, Ph.D. Principal Scientist in DMPK Service Department of WuXi AppTec, has 5 years of experience in drug metabolite profiling and identification with strong expertise in macromolecular LC-MS analysis.
Dr. Guoxiang Huang, Ph.D. in Synthetic Organic Chemistry, Senior Research Fellow in DMPK Service Department of WuXi AppTec, has over 22 years of drug discovery experience in medicinal chemistry and drug metabolite profiling and identification and extensive experience in the preclinical development of drugs. He successfully supported many new drug IND applications globally.
Ms. Weiqun Cao, Director in DMPK Service Department of WuXi AppTec, has over 17 years of experience in bioanalysis, metabolite profiling and identification of drugs.
Filed Under: Drug Discovery and Development





Tell Us What You Think!
You must be logged in to post a comment.