Editor’s Note: This technical paper is adapted from a webinar sponsored by Dassault Systèmes and presented by Advantage Business Marketing. The webinar featured Rakesh Dixit, PhD, as the speaker, who is an expert in the field of biologics from Medimmune and who presented his insights on
the future of the transformative biologics market and helped navigate the webinar discussion. Laura Panjwani, the R&D Magazine Editor, served as the moderator.The full webcast can be viewed here: https://event.webcasts.com/starthere.jsp?ei=1192416&tp_key=1cd32f57f1
The biopharmaceutical market is a rapidly growing class of therapeutics, showing significant potential in oncology, diabetes and other disease areas. Unlike conventional chemically synthesized pharmaceuticals, biopharmaceuticals—also known as biologics—are derived from living organisms, typically using biotechnology.
Examples of biologics include hormones, blood products, cytokines, monoclonal antibodies (mAbs), and vaccines, as well as gene transfer, cell therapy and tissue engineered products.
There are more than 300 mAbs, more than 250 vaccines, and more than 100 other biologics— including cell and gene therapies—currently in clinical development. The global biologics market is expected to reach around $291 billion in 2020 and by 2022, 50 percent of the pharmaceutical market share is expected to be in biologics.
But the future of biologics won’t be focused solely on the discovery of new therapeutics. There is also a significant market for biosimilars, biologic drugs that demonstrate high similarity to an already approved biologic reference drug, and can in turn serve as an alternative to it. Biosimilars are different than generics, which are synthetic chemical copies of their reference drugs and are identical in active ingredients, strength, dosage form and route of administration.
Because biologics are made with living cells and no two molecules can be exactly the same, making a copy of a biologic is a much more involved and expensive process than creating a generic drug. There will always be very subtle differences between the biosimilar and its reference biologic.
There are significant benefits to biosimilars. Biosimilars are a more cost effective way to manufacture biologics, increasing the affordability of life-saving biologics to patients.
They could also increase worldwide regulatory approvals, including with the U.S. FDA. The Patient Protection and Affordable Care Act was signed into law by President Barack Obama on March 23, 2010, amending the Public Health Service Act to create an abbreviated approval pathway for biosimilars that are considered “highly similar” with an FDA-approved biological product.
It is expected that by 2030 the biosimilar market will be a greater than a $240 billion opportunity, as patents on major biologics continue to expire.
To be approved, biosimilars need to prove a high degree of similarity in biophysical properties, safety and effectiveness to the marketed reference product. The agent must demonstrate no meaningful difference in immunogenicity and pharmacokinetic/ pharmacodynamic (PK/PD) outcomes in a well-designed clinical trial. However, lower immunogenicity and better safety are acceptable.
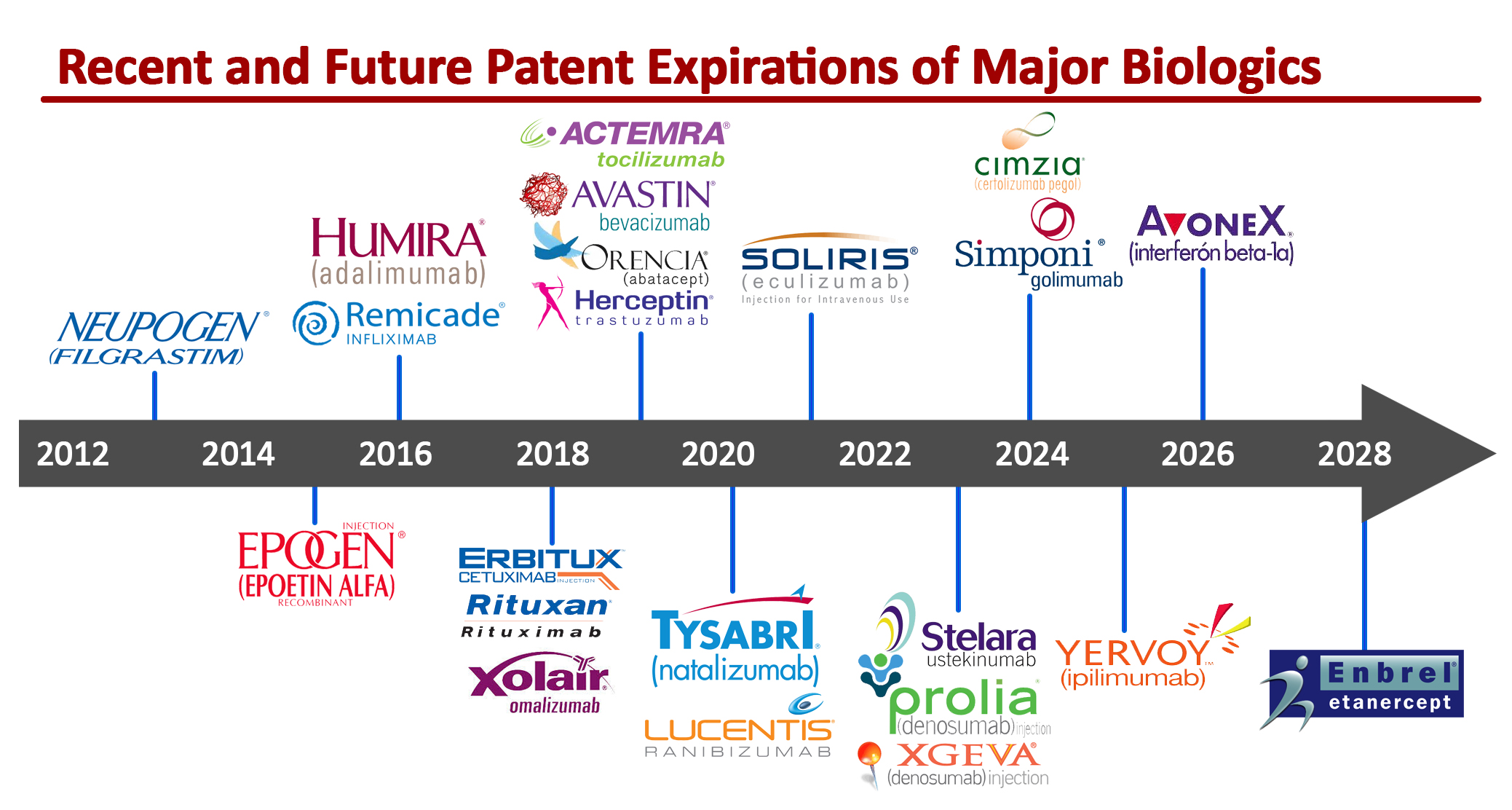
Sources: EvaluatePharma, Cortells
Challenges facing the biosimilar market
Despite its potential in cost savings, the biosimilar market still faces hurdles. There are gaps in knowledge and understanding of biosimilars among the public, and that can lead to challenges with physicians and patients’ perception of the safety and efficacy of the product.
Worldwide there is also a high variability in regulations regarding the approval of biosimilars. In Europe, the regulatory pathway and litigation procedures are much clearer and more defined. In the U.S., however, there are very broad IP and patent laws, which can make patent infringements a major obstacle in effective marketing. This can impact patient access and increase cost.
Health plans and pharmacy benefit managers (PBMs) can also contribute to increased costs, as some require high rebates that block biosimilar access and uptake. Finally, multiple companies also often compete to make the same biosimilar of a biological drug that has recently come off patent, causing stiff competition across the market.
Overcoming these obstacles
To stand out from the competition, companies that make biosimilar products can consider lowering prices or offering high rebates to PBMs. However, this approach is best suited in developing countries with high price sensitivity. In more established markets for biologics, such as the U.S., Europe and Japan, pricing alone isn’t sufficient, as there typically isn’t significant price variety among similar therapeutics.
It is also possible to attempt to differentiate the molecule itself by providing better tolerability. However, regulatory agencies will not typically allow bio superiority to be claimed on a label on a biosimilar product.
One potentially effective way to differentiate a biosimilar is through a novel delivery device or container closure system, which improves convenience, ease of use or patient acceptability of the therapeutic from its reference product. Some design differences in the delivery device or container closure system used with the proposed biosimilar product also may be acceptable for regulatory approval of a biosimilar.
For a proposed biosimilar product in a different delivery device or container closure system, the presentation must be shown to be compatible for use with the final formulation of the biological product through appropriate studies including, for example, extractable/leachable studies and stability studies.
For certain design differences in the delivery device or container closure system, performance testing and a human factors study may be needed.
“Biobetters”
Enhancing a biosimilar with an improved or more convenient delivery system is not the only way to differentiate a product.
Highly differentiated biosuperior drugs, known as “Me-Betters” (after the term “Me-Too” drugs used to describe two highly similar therapeutics) are needed to serve patients who have increasingly become resistant to current therapies/standard of care.
These “Me-Betters” or “biobetters,” are new biologics based on an existing approved biologic. Biobetters can deliver the mechanism of action of potency improvements, enhanced half-life, better safety and immunogenicity, and better and broader efficacy.
These biobetters may serve high unmet medical needs more rapidly than a novel therapeutic, as they often receive shorter review and more rapid approval with better reimbursement.
There are numerous biosimilars and biobetters in development today.

Biobetter strategy
Biopharmaceutical companies have focused on several strategies to develop biobetters. They are only working with molecules that have mechanisms of action (MOA) that are clinically proven or have a proof-of-efficacy that has been established and where addition values can be gained. They are focusing on areas where there are unmet medical needs within a known class of agents, where current drugs or their biosimilars do not already serve well.
They aim to create biobetters where current agents are inadequate to treat refractory patients, relapsed patients, or those that have inconvenient dosing systems or safety concerns. To do this, they are focusing on application of best science and antibody technologies to create highly differentiated and potent biologics within the same general MOA as already established agents.
Pros and cons of developing biobetters
There are several positive outcomes from creating a highly differentiated biosuperior drug. Unlike with a biosimilar, there is generally no need to wait for patents to expire because all biobetters are treated as new molecular entries from a regulatory perspective.
Despite these benefits, developing biobetters does come with challenges. As compared to biosimilars, the regulatory process will be longer and more expensive, as the agent is treated as an entirely new entry. As a result, clinical development cost may not be too dissimilar to innovative drug development.
Biobetters also face fierce challenges for demonstrating superiority in efficacy or safety against established biologics and market leaders, and unless the benefits are superior to biosimilars, the higher costs of biobetters may be questioned. It can be complex to establish biosuperority, and not every attempt at doing so will be successful.

Credit: Medimmune
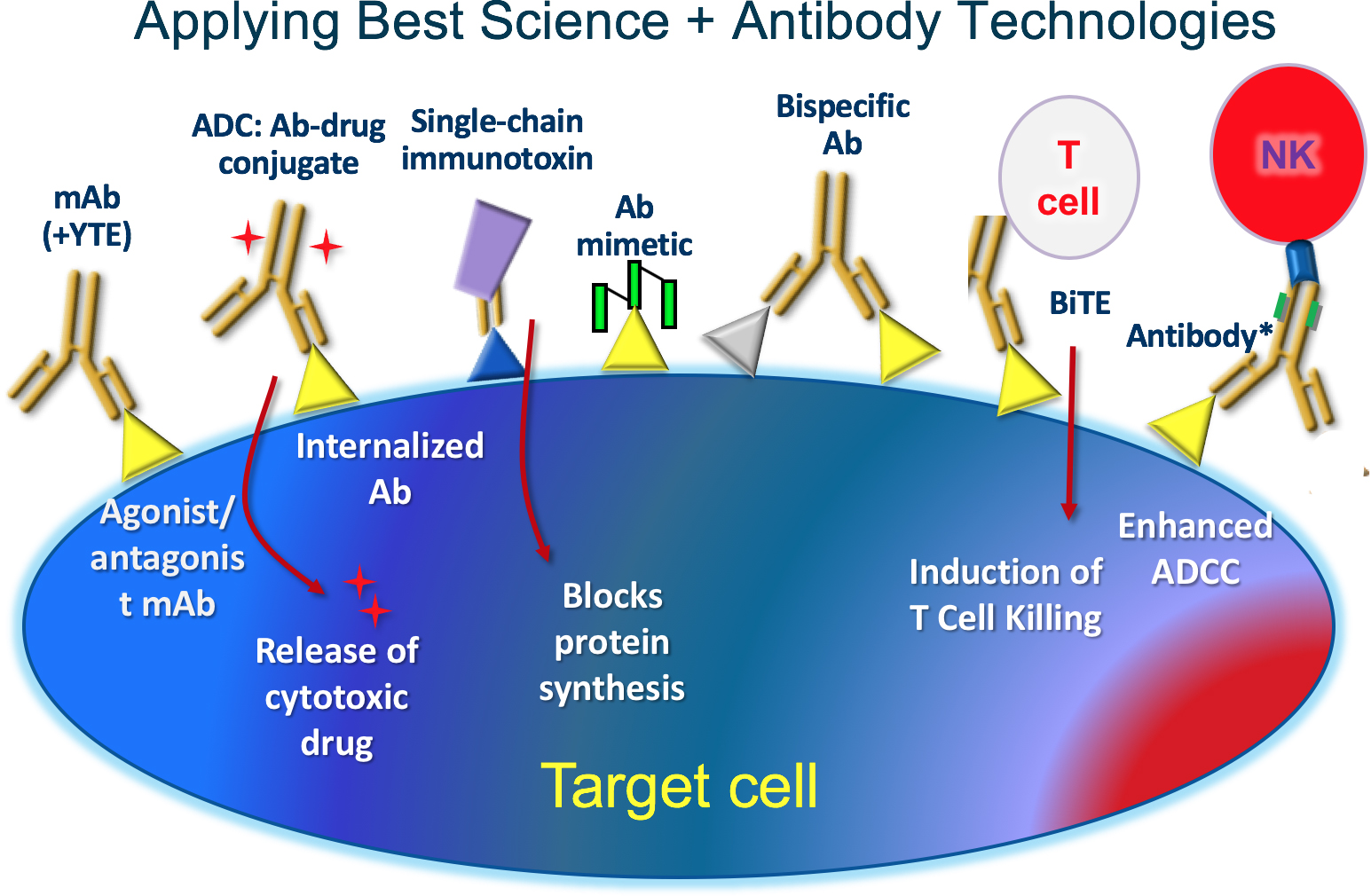
Credit: Medimmune
One example of an extremely successful biobetter is Humira (adalimumab), an immunosuppressive drug used to treat arthritis, plaque psoriasis, ankylosing spondylitis, Crohn’s disease, and ulcerative colitis and other diseases. Humira works by binding to tumor necrosis factor-alpha (TNFα), which normally binds to TNFα receptors, leading to the inflammatory response of autoimmune diseases.
Humira was the third TNF inhibitor, after infliximab and etanercept, to be approved in the U.S. A “biobetter” of the two agents, it was constructed from a fully human monoclonal antibody, which was an improvement over infliximab, a mouse-human chimeric antibody, and etanercept, a TNF receptor- IgG fusion protein.
Humira resulted from collaboration between BASF Bioresearch Corporation and Cambridge Antibody Technology (CAT), which became MedImmune in 2007. The key to Humira’s discovery was the “phage display” method discovered at MedImmune. As of 2017, the drug has made more than 18 billion in the U.S.
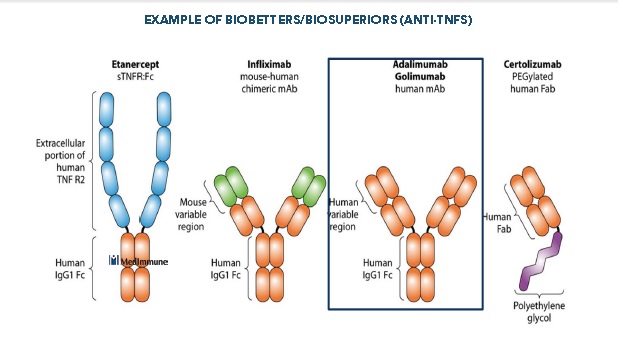
Credit: Medimmune
Conclusion
Biosimilars have tremendous potential to reduce the high cost of biologics and increase the affordability of biologics medicines. However, there are still barriers to the market’s success on the large scale, including patent limitations and legal challenges, a fractionated market, high discount expectations from PBMs and physician and patient acceptance.
For innovation companies, highly differentiated and technologically advanced biobetters offer the greatest potential to mitigate the risks associated with the introduction of biosimilars, as they can be approved before the patent expires and have blockbuster potential. However, strategy is key, as long development time, high cost, and uncertainty in biosuperiority over existing drugs can hinder the success of a biobetter agent.

Filed Under: Drug Discovery



