Welcome to Drug Discovery Spotlight — we’ve rounded up this week’s top news, trending headlines, and all-around interesting reads.
 Zika Is Linked to Microcephaly, Health Agencies Confirm
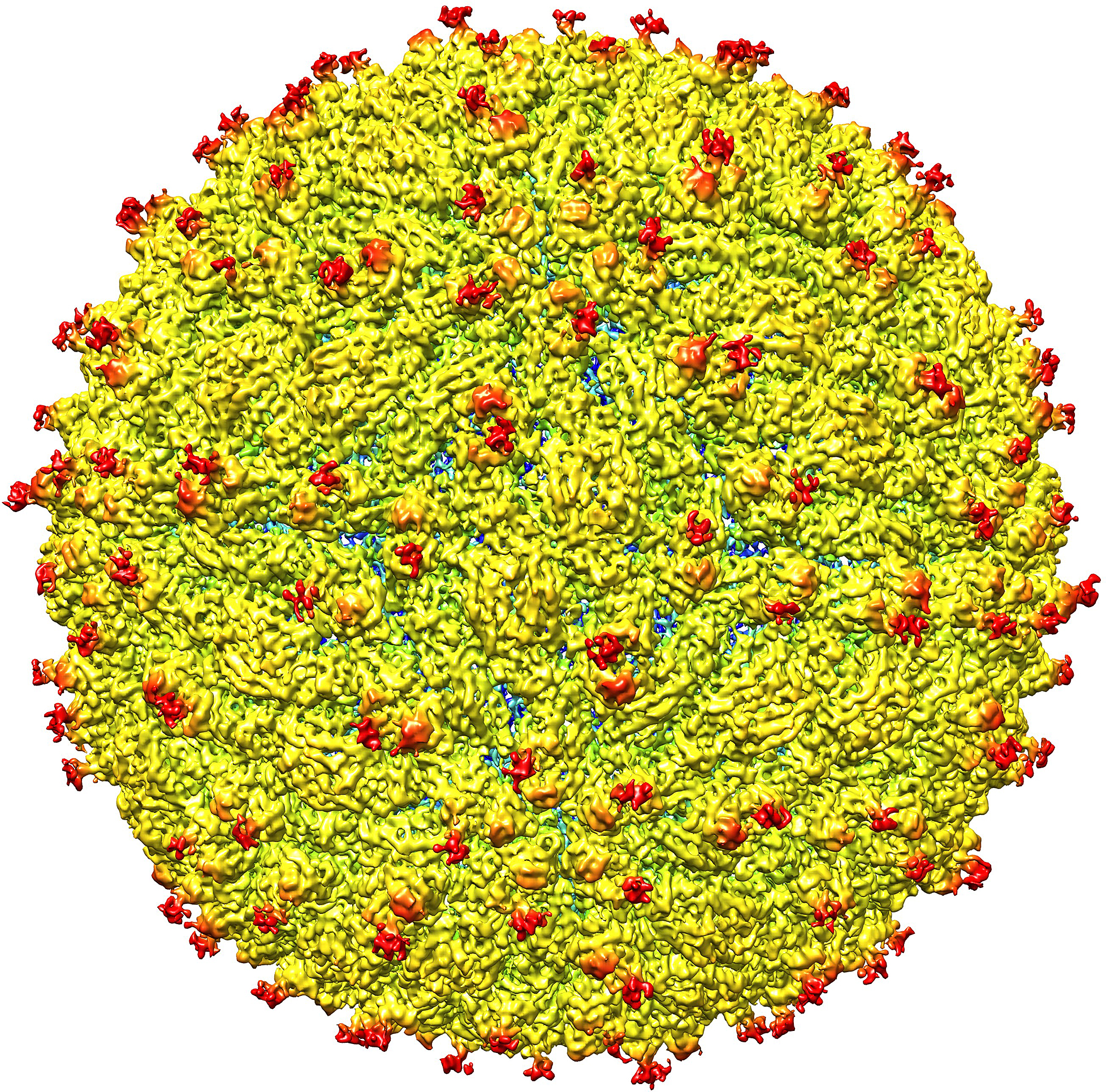
Zika Is Linked to Microcephaly, Health Agencies Confirm
The World Health Organization has confirmed in a scientific consensus that the Zika virus is linked with microcephaly, the condition in which babies are born with small heads and brain damage. It’s been thought for some time that Zika and microcephaly are associated, but this week the WHO and Centers for Disease Control and Prevention have agreed that there is enough evidence to officially say that the virus and condition are connected. This week a report, published in Science, announced that an image of the 3D structure of the Zika virus has been revealed. Its spherical structure is said to resemble other viruses, like the dengue virus, but it also contains differences, such that its outer shell is not similar. The finding has implications in assisting scientists in preventing the virus’s transmission. Scientists from ‘rival disciplines’ are also collaborating in an attempt to assemble a state-of-the-art DNA map of a mosquito’s genetic code.
FDA Eases Requirements on Abortion Pill Label
The FDA on Wednesday eased up on requirements for taking a medication that induces abortion, reviving a contentious debate. The agency said its actions were strictly based on medical science. The New York Times reported, “The change brings the directions for taking the drug, mifepristone, in line with what has become standard medical practice in most states: reducing the dosage to 200 milligrams from 600 milligrams, decreasing the number of visits a woman must make to a doctor to two from three, and extending the period when she can take the pill to 10 weeks of pregnancy from seven weeks.”
‘We Thought We Were Curing People’: Hope Dims Ovarian Cancer Therapy Fails Test
Gynecological oncologists across the country were dismayed to learn earlier this week that a major study of a promising treatment for ovarian cancer failed. (Fewer than half of ovarian cancer patients survive five years). The study showed that intraperitoneal (IP) chemotherapy was no more effective than traditional treatments. “You could have heard a pin drop,” said one doctor who was in the filled conference room when the results were announced. Read more at STAT.
Chinese Market Offers New Life to Many Drugs
Prescription medications that have failed to make it to market in the U.S. and other countries are being reevaluated in China, as in the example of BMS’s licensure of a first-line liver-cancer drug called brivanib to a Chinese startup. The Wall Street Journal, though, raises the question: Has China become a dumping ground for inferior drugs?
Filed Under: Drug Discovery




