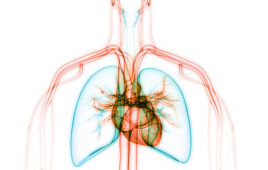
Idiopathic Pulmonary Fibrosis (IPF), a devastating lung disease affecting millions with increasing incidence, may have a new treatment hope thanks to a novel inhibitor of TNIK, a kinase newly implicated in fibrosis, identified using generative AI drug discovery platforms in just 18 months. Researchers at Insilico Medicine, along with international collaborators, harnessed the power of…
Fixed-dose macitentan-tadalafil polypill outperforms monotherapies in PAH
Pulmonary arterial hypertension (PAH) is a relatively rare disorder, affecting roughly 15 to 50 people per million within the U.S. and Europe. Treating it with a single therapy can be challenging, and medication adherence often presents a common hurdle for the condition. Combination therapy has thus become a widely used option in the treatment of…
CEO: Insilico on how AI can ‘imagine the perfect molecules’ for drug targets
Insilico Medicine, an AI-based biotech startup, announced details of their first AI-designed drug candidate to enter human clinical trials. INS018_055 is an experimental treatment for idiopathic pulmonary fibrosis, a rare lung disease. Through their Insilico AI-driven drug discovery platform, they discovered and designed INS018_055 in just 30 months, significantly faster than the industry average. Phase…
Insilico’s AI-discovered INS018_055 graduates to phase 2
Roughly a year ago, Insilico Medicine announced that it had dosed the first patient in a phase 1 study of INS018_055, an AI-discovered, first-in-class small molecule inhibitor. Now, the company has progressed to the next stage, launching a phase 2 study for the drug candidate. Insilico, a founding member of NVIDIA Inception, developed its AI…
FDA advisory committee backs AstraZeneca’s albuterol/budesonide for asthma
FDA’s Pulmonary-Allergy Drugs Advisory Committee (PADAC) voted 16 to 1 in favor of the albuterol/budesonide (PT027) from AstraZeneca (Nasdaq:AZN) to treat asthma in people at least 18 years old. The voting proposition asked whether the data supported a favorable benefit-risk assessment for various age groups. The advisory committee did not endorse albuterol/budesonide for patients 12…
Boehringer Ingelheim launches Phase 3 study for novel IPF therapy
Currently, there are only two FDA-approved drugs for idiopathic pulmonary fibrosis (IPF) — Ofev (nintedanib) from Boehringer Ingelheim and Esbriet (pirfenidone) from Genentech. Now, Boehringer Ingelheim is announcing the launch of the global FIBRONEER-IPF study focused on BI 1015550, an experimental phosphodiesterase 4B (PDE4B) inhibitor, in idiopathic pulmonary fibrosis (IPF). The study is part of…
Pulmonary Fibrosis Foundation’s latest registry offers research opportunities
The Pulmonary Fibrosis Foundation (PFF) has launched the PFF Community Registry to facilitate research on pulmonary fibrosis (PF) and interstitial lung disease (ILD). The PFF Community Registry collects self-reported data from patients, nonprofessional caregivers and biological family members via a secure online portal. Participation is voluntary. “There’s a research opportunity to see if there are any differences…
Janssen reports positive clinical trial data for its Xarelto to treat PAD effects
Johnson & Johnson’s (NYSE:JNJ) Janssen Pharmaceutical Companies recently announced analysis data on the benefits of Xarelto. The Phase 3 Voyager PAD clinical trial evaluated the Xarelto (rivaroxaban) vascular dose (2.5 mg twice daily plus aspirin 100 mg once daily) in reducing severe vascular events in patients with peripheral artery disease (PAD) after lower-extremity revascularization (LER).…
MannKind shares slide 22% as FDA delays review of Tyvaso DPI new drug application
Inhalable insulin specialist MannKind Corp. (NASDAQ:MNKD) announced that the FDA is extending its deadline to review United Therapeutics’ (NASDAQ:UTHR) new drug application (NDA) for Tyvaso DPI from February to May. MannKind is developing Tyvaso DPI (treprostinil) in collaboration with United Therapeutics. The drug candidate Tyvaso DPI would potentially treat pulmonary arterial hypertension and pulmonary hypertension…
4 potential pulmonary fibrosis therapies to watch
The drug development landscape in idiopathic pulmonary fibrosis is changing rapidly. “There are probably 30 or 40 different compounds in development,” said Dr. Toby Maher, a presenter at the Pulmonary Fibrosis Foundation (PFF) Summit held last month. Of those, a handful have reached Phase 2 and Phase 3 clinical trials. Here, we highlight a handful…
TFF Pharmaceuticals sees promise in Phase 1 trial of inhaled tacrolimus powder for lung transplant
The biopharma TFF Pharmaceuticals (NSDQ:TFFP) has announced that its inhalation powder of the immunosuppressive drug tacrolimus was well tolerated in a Phase 1 trial in lung transplant patients. Tacrolimus is a traditional option for preventing rejection of a transplanted organ, albeit in tablet or capsule form. It is common for lung transplant recipients to have…
Why Aria Pharmaceuticals is upbeat about two novel IPF treatment candidates
Small molecule drug developer Aria Pharmaceuticals (Palo Alto, Calif.) recently revealed that two investigational treatments for idiopathic pulmonary fibrosis (IPF), TXR-1002 and TXR-1007, demonstrated promising results in preclinical research. There are currently only two FDA-approved treatments for IPF on the market — nintedanib (Ofev) from Boehringer Ingelheim and pirfenidone (Esbriet) from Roche. While those introductions…
Why pulmonary fibrosis deserves more attention
This September is Pulmonary Fibrosis Awareness Month, which the Pulmonary Fibrosis Foundation (PFF) and allies have launched to educate the public about the disease family. Involving scarring of the lungs, pulmonary fibrosis gradually robs the breath from patients it affects. Some 250,000 Americans live with idiopathic pulmonary fibrosis, according to PFF. At present, two drugs…
Janssen’s pulmonary hypertension division leader is upbeat about the future of PH treatment
A few decades ago, an HIV diagnosis was akin to a death sentence. But with proper treatment including antiretroviral therapy, HIV patients can often expect to have a normal life expectancy. David Jimenez, former president of Johnson & Johnson’s Janssen Infectious Diseases, is confident that similar breakthroughs can happen in pulmonary hypertension. “We’re not there…