Boston Children’s Hospital announced today that it partnered with ElevateBio for a five-year cell and gene therapy advancement program. The collaborative agreement will seek to advance cell and gene therapy programs originating out of Boston Children’s Hospital with an introduction to Boston Children’s translational research capabilities. Get the full story at our sister site, Drug Delivery Business…
Sanofi to spend nearly half a billion dollars a year on mRNA vaccine research
Sanofi (NYSE:SNY) announced today that it plans to invest approximately $476.3 million (€400 million) per year on mRNA vaccine development. Paris-based Sanofi’s intends for its annual investment to go toward a first-of-its-kind vaccine “mRNA Center of Excellence” to accelerate the development and delivery of next-generation vaccines. Approximately 400 employees will integrate end-to-end mRNA vaccine capabilities…
San Diego company aiming to target glioblastoma with nanotechnology-based approach
The emerging biopharmaceutical company Global Cancer Technology (GCT; San Diego) is developing a novel x-ray–activated modality for drug delivery to treat glioblastoma, which remains stubbornly difficult to treat. GCT has a licensing agreement with the University of California, San Diego (UCSD) for a drug delivery platform that unites a nanocrystal to a prodrug that can be…
Biden administration invests $3B to accelerate development of next-gen COVID-19 treatments
President Joe Biden’s administration will spend $3 billion in American Rescue Plan money to accelerate the discovery, development and manufacturing of antiviral medicines against COVID-19, HHS announced today. Not only will the Antiviral Program for Pandemics seek to boost the availability of medicines to prevent serious COVID-19 illness and save lives, but it will pursue sustainable…
Bayer moves forward with cell and gene therapies for Parkinson’s disease
Bayer announced today that its BlueRock Therapeutics subsidiary successfully administered the first dose of its Parkinson’s disease therapies. Berlin-based Bayer said in a news release that BlueRock successfully administered the dose of its investigational pluripotent stem cell-derived dopaminergic neruons, called DA01, to a Parkinson’s disease patient in an open-label Phase 1 clinical safety and tolerability…
Moderna seeks full FDA approval for its COVID-19 vaccine
Moderna (Nasdaq:MRNA) announced today that it submitted for full FDA approval of its mRNA COVID-19 vaccine for individuals 18 years of age and older. Cambridge, Mass.-based Moderna initiated the rolling submission process with the FDA for a Biologics License Application (BLA) to license its mRNA COVID-19 vaccine, according to a news release. The move…
EU won’t renew J&J, AstraZeneca vaccine contracts, report says
Reports from Italy claim that the European Union Commission won’t renew COVID-19 vaccine contracts next year with AstraZeneca and Johnson & Johnson. According to Reuters, Italian outlet La Stampa reported that sources from the Italian health ministry have said the commission does not want to pursue contract renewals with those two companies once those encompassing the…
Pfizer to boost vaccine production for U.S. by 10%
Pfizer will deliver 10% more of its COVID-19 vaccine to the U.S. by the end of May than originally planned, its CEO tweeted yesterday. The tweet follows the federal government’s decision to recommend a temporary halt in the distribution of Johnson & Johnson’s single-dose vaccine, which has been tied to rare instances of blood clot…
Feds halt J&J COVID-19 vaccine due to blood clot concern
The FDA and CDC issued a joint statement today recommending the pause in the administration of the Johnson & Johnson COVID-19 vaccine. As of yesterday, more than 6.8 million doses of the J&J single-dose vaccine had been administered in the U.S. Among recipients of those doses, six cases of a rare and severe type of blood…
Intravacc touts preclinical, non-human results for COVID-19 nasal spray vaccine
Intravacc is today touting positive pre-clinical results of its SARS-CoV-2 nasal spray vaccine candidate in non-human subjects. Bilthoven, Netherlands-based Intravacc’s SARS-CoV-2 outer membrane vesicle (OMV)-based recombinant spike protein (rSp) nasal spray candidate was observed in four groups of mice and four groups of hamsters, all of which received two intranasal immunizations on day one and day…
EU allows Pfizer/BioNTech COVID-19 vaccine to be stored at higher temperatures
Pfizer and BioNTech announced today that European authorities approved storage of their COVID-19 vaccine at a new temperature. Based on data showing stability in standard pharmaceutical freezers, the European Medicines Agency (EMA) approved storage of the vaccine at -25°C to -15°C for a total of two weeks. The FDA’s requirements include two-week storage at the same temperatures,…
FDA approves oral multiple sclerosis treatment from J&J’s Janssen unit
Johnson & Johnson (NYSE:JNJ) unit Janssen Pharmaceutical announced today that it received FDA approval for its Ponvory treatment. Ponvory (ponesimod) is a once-daily, oral selective sphingosine-1-phosphate receptor 1 (S1P1) modulator for treating adults with relapsing forms of multiple sclerosis (MS). It’s areas of treatment include clinically isolated syndrome, relapsing-remitting disease and active secondary progressive disease,…
Oramed forms joint venture to develop oral COVID-19 vaccines with POD delivery tech
Oramed Pharmaceuticals (NSDQ:ORMP) announced today that it is forming a joint venture to develop novel oral COVID-19 vaccines. New York-based Oramed entered into definitive agreements to form Oravax Medical, which is based on Oramed’s proprietary POD (protein oral delivery) technology and Premas Biotech’s novel vaccine technology, according to a news release. Get the full story…
HRS Heat Exchangers is helping to support COVID-19 vaccine production: Here’s how
HRS Heat Exchangers announced that it received orders for pharmaceutical heat exchangers for use with the manufacturing of COVID-19 vaccines. Orders were placed by subcontractors working with Pfizer and Moderna, which both have vaccines authorized in the U.S., and Oxford/AstraZeneca, which has been approved across Europe but not yet in the U.S. The orders are…
Denmark is investigating reports of blood clots after AstraZeneca COVID-19 vaccinations
The Danish Medicines Agency announced that it is investigating the AstraZeneca COVID-19 vaccine after reports of blood clots. In collaboration with the European Medicines Agency, the Danish Medicines Agency launched the investigation as it considers there to be a reason to look into the circumstances further after cases in which citizens developed blood clots shortly…
Report: Biden to buy 100M additional doses of J&J COVID-19 vaccine
President Joe Biden is reportedly set to announce plans to purchase an additional 100 million doses of Johnson & Johnson’s (NYSE:JNJ) COVID-19 vaccine. An NBC News report said that Biden is set to announce the purchase of the single-dose vaccines on Wednesday, according to two officials from his administration. The purchase of doses from Johnson &…
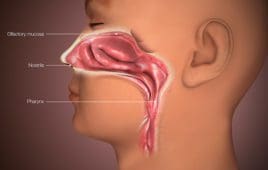
How intranasal vaccines can confer immunity where it matters
The COVID-19 vaccine landscape is gradually becoming more diverse. And it could grow more so, as several companies work on intranasal vaccines. Intranasal vaccines offer potential advantages over traditional intramuscular vaccines. They can stimulate immunoglobulin A production, which can avert infection, as a recent Scientific American article noted. [Related: Intranasal COVID-19 vaccines could be on the…
Merck moving to Phase 2 study of drug-eluting implant to prevent HIV infection
Merck (NYSE:MRK) today announced positive results from a Phase 1 study of its subdermal drug-eluting implant for preventing HIV-1 infection. The investigational implant has the potential for extended administration of islatravir for pre-exposure prophylaxis (PrEP) of HIV-1 infection, according to a news release. Get the full story at our sister site, Drug Delivery Business News.
Report: Biden to announce partnership for Merck to make J&J’s COVID-19 vaccine
President Joe Biden is reportedly set to announce a partnership in which Merck (NYSE:MRK) will make Johnson & Johnson’s (NYSE:JNJ) recently approved COVID-19 vaccine. The Washington Post reported that the agreement between the two competitors will boost the supply of the single-dose vaccine from J&J’s Janssen subsidiary, which received FDA emergency use authorization last week. Get the full story at…
Kala Pharmaceuticals slides on missed Q4 projections
Kala Pharmaceuticals (NSDQ:KALA) shares dipped this morning on fourth-quarter results that came up short of the consensus forecast. The Watertown, Mass.-based company posted losses of -$31.1 million, or -55¢ per share, on sales of $2.2 million for the three months ended Dec. 31, 2020, for a more than $10 million bottom-line slide on sales growth of…
J&J submits EUL application to WHO for single-shot COVID-19 vaccine
Johnson & Johnson (NYSE:JNJ) announced today that it submitted for emergency use listing (EUL) to the World Health Organization (WHO) for its COVID-19 vaccine. New Brunswick, N.J.-based J&J’s Janssen subsidiary’s single-dose COVID-19 vaccine candidate was submitted for EUL with a data package including interim efficacy and safety results from the Phase 3 Ensemble clinical trial, which…
Report: Pfizer speeding up vaccine production
Pfizer (NYSE:PFE) is reportedly expecting to trim the time needed to produce COVID-19 vaccine batches to speed up production. The company, which along with BioNTech (NSDQ:BNTX) developed one of two COVID-19 vaccines currently authorized in the U.S., is slated to cut the amount of time it takes to produce a batch of the vaccine from 110…
Facial swelling and COVID-19 vaccines: 4 facts
Patients with dermal fillers have a small potential risk of developing facial swelling after obtaining mRNA vaccines. To date, the FDA has noted the problem in three clinical trial recipients of the COVID-19 vaccine from Moderna (NSDQ:MRNA). The agency hasn’t observed facial swelling in the Phase 3 clinical trial for the COVID-19 vaccine from Pfizer…
Merck inks supply deal with U.S. for investigational COVID-19 therapy
Merck (NYSE:MRK) has agreed to supply the U.S. government with the biological therapeutic CD24Fc if FDA authorizes the use of the drug. The drug played a central role in Merck’s decision to acquire privately held OncoImmune, which had touted CD24Fc as its “lead compound.” Merck plans on renaming the CD24Fc therapy MK-7110. (We recently profiled the…
Ensuring safe and effective distribution of COVID-19 vaccines
By Stewart Eisenhart, Emergo Group As governments, industry and other entities begin COVID-19 vaccine distribution efforts worldwide, cold chain management has emerged as a crucial factor for ensuring vaccine safety and effectiveness. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the author’s only and do…